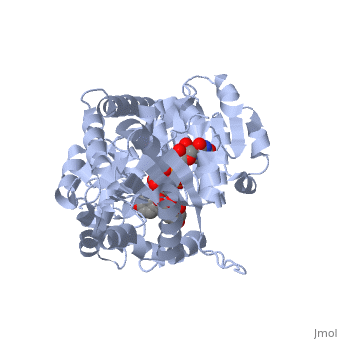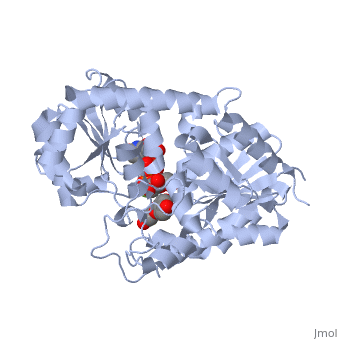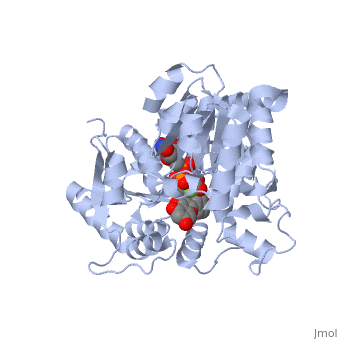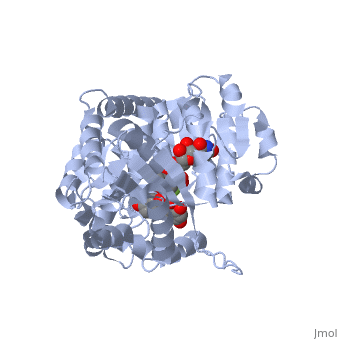
Vitis vinifera Flavonoid 3-O-Glucosyltransferase (Vv3GT)
From Proteopedia
(Difference between revisions)
m (adding pages to the "Pages with quizzes" category) |
|||
| (One intermediate revision not shown.) | |||
| Line 1: | Line 1: | ||
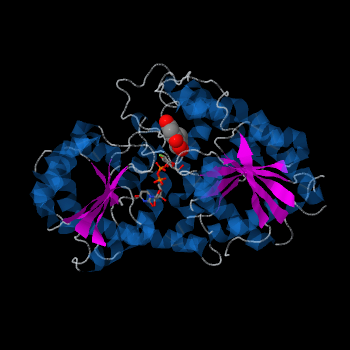
| - | <StructureSection load='2c1z' size='340' side='right' caption=' | + | <StructureSection load='2c1z' size='340' side='right' caption='Flavonoid 3-O-glucosyltransferase complex with UDP-glucose derivative and kaempherol (PDB code [[2c1z]])' scene=''> |
''Vitis vinifera'' '''Flavonoid 3-O-Glucosyltransferase''' (Vv3GT) is involved in the modification of grape [http://en.wikipedia.org/wiki/Anthocyanin anthocyanins] (a plant pigment) and thus could affect their water solubility and color stability <ref>PMID:21443631</ref>. The addition of a sugar molecule on the anthocyanin is a preliminary step to its transport to the cell vacuole. The transfer to the vacuole is important for the pigment accumulation. The anthocyanin accumulation plays a significant role in quality of agricultural produce, as it affects fruit color and its health benefits as a natural antioxidant. This enzyme affects the quality of both table grapes and wine grapes. | ''Vitis vinifera'' '''Flavonoid 3-O-Glucosyltransferase''' (Vv3GT) is involved in the modification of grape [http://en.wikipedia.org/wiki/Anthocyanin anthocyanins] (a plant pigment) and thus could affect their water solubility and color stability <ref>PMID:21443631</ref>. The addition of a sugar molecule on the anthocyanin is a preliminary step to its transport to the cell vacuole. The transfer to the vacuole is important for the pigment accumulation. The anthocyanin accumulation plays a significant role in quality of agricultural produce, as it affects fruit color and its health benefits as a natural antioxidant. This enzyme affects the quality of both table grapes and wine grapes. | ||
== Introduction == | == Introduction == | ||
| Line 107: | Line 107: | ||
*[http://en.wikipedia.org/wiki/Glycosyltransferase Glycosyltransferases in WikipediA] | *[http://en.wikipedia.org/wiki/Glycosyltransferase Glycosyltransferases in WikipediA] | ||
*[http://www.cazy.org CAZy database] | *[http://www.cazy.org CAZy database] | ||
| + | |||
| + | [[Category:Pages with quizzes]] | ||
Current revision
| |||||||||||
References
- ↑ Yonekura-Sakakibara K, Hanada K. An evolutionary view of functional diversity in family 1 glycosyltransferases. Plant J. 2011 Apr;66(1):182-93. doi: 10.1111/j.1365-313X.2011.04493.x. PMID:21443631 doi:http://dx.doi.org/10.1111/j.1365-313X.2011.04493.x
- ↑ Frydman A, Weisshaus O, Bar-Peled M, Huhman DV, Sumner LW, Marin FR, Lewinsohn E, Fluhr R, Gressel J, Eyal Y. Citrus fruit bitter flavors: isolation and functional characterization of the gene Cm1,2RhaT encoding a 1,2 rhamnosyltransferase, a key enzyme in the biosynthesis of the bitter flavonoids of citrus. Plant J. 2004 Oct;40(1):88-100. PMID:15361143 doi:http://dx.doi.org/10.1111/j.1365-313X.2004.02193.x
- ↑ Osmani SA, Bak S, Moller BL. Substrate specificity of plant UDP-dependent glycosyltransferases predicted from crystal structures and homology modeling. Phytochemistry. 2009 Feb;70(3):325-47. doi: 10.1016/j.phytochem.2008.12.009. Epub, 2009 Feb 13. PMID:19217634 doi:http://dx.doi.org/10.1016/j.phytochem.2008.12.009
- ↑ Osmani SA, Bak S, Moller BL. Substrate specificity of plant UDP-dependent glycosyltransferases predicted from crystal structures and homology modeling. Phytochemistry. 2009 Feb;70(3):325-47. doi: 10.1016/j.phytochem.2008.12.009. Epub, 2009 Feb 13. PMID:19217634 doi:http://dx.doi.org/10.1016/j.phytochem.2008.12.009
- ↑ Offen W, Martinez-Fleites C, Yang M, Kiat-Lim E, Davis BG, Tarling CA, Ford CM, Bowles DJ, Davies GJ. Structure of a flavonoid glucosyltransferase reveals the basis for plant natural product modification. EMBO J. 2006 Mar 22;25(6):1396-405. Epub 2006 Feb 16. PMID:16482224