|
|
| (25 intermediate revisions not shown.) |
| Line 1: |
Line 1: |
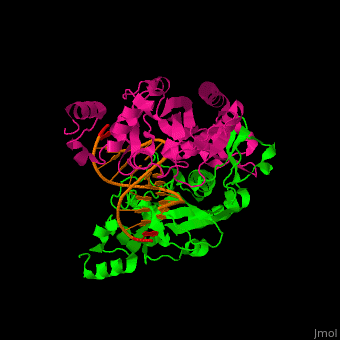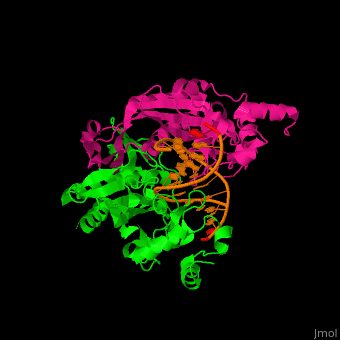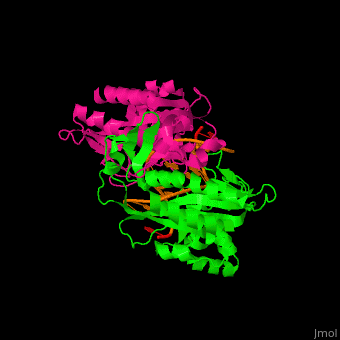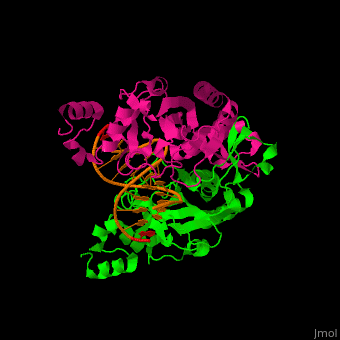
| - | <StructureSection load='1rva' size='350' side='right' scene='49/496318/Cv/1' caption='E. coli EcoRV endonuclease dimer (magenta, green) complex with DNA, (PDB code [[1rva]])'> | + | <StructureSection load='' size='350' side='right' scene='49/496318/Cv/1' caption='E. coli EcoRV endonuclease dimer (magenta, green) complex with DNA, (PDB code [[1rva]])'> |
| | | | |
| | == Function == | | == Function == |
| | | | |
| | '''Endonuclease''' (ENN) cleaves phosphodiester bond within polynucleotide chain. ENN cleaves DNA at a restriction site which is usually a 6-nucleotide palindrome. ENN is restriction site–specific. Various types of ENN differ by their mechanism of action. ENN is used in genetic engineering to make recombinant DNA. ENN requires a restriction site and a cleavage pattern. '''ENN-I''' operates on DNA with separate restriction site and cleavage pattern, while '''ENN-II''' operates on overlapping restriction site and cleavage pattern. Some ENNs are encoded within introns thus facilitating their mobility. These ENNs or inteins are designated I-ENN<ref>PMID:12483517</ref>.<br /> | | '''Endonuclease''' (ENN) cleaves phosphodiester bond within polynucleotide chain. ENN cleaves DNA at a restriction site which is usually a 6-nucleotide palindrome. ENN is restriction site–specific. Various types of ENN differ by their mechanism of action. ENN is used in genetic engineering to make recombinant DNA. ENN requires a restriction site and a cleavage pattern. '''ENN-I''' operates on DNA with separate restriction site and cleavage pattern, while '''ENN-II''' operates on overlapping restriction site and cleavage pattern. Some ENNs are encoded within introns thus facilitating their mobility. These ENNs or inteins are designated I-ENN<ref>PMID:12483517</ref>.<br /> |
| - | The '''Cas ENN''' proteins are part of CRISPR/Cas prokaryotic immune system which confers protection from foreign genetic elements like viruses. The CRISPR (Custered Regularly Interspersed Short Palindromic Repeats) are DNA loci which are found in ca. 40% of the bacteria. The CRISPR/Cas system is being used lately as gene editing tool<ref>PMID:20056882</ref>. For more details see [[Cas9]].<br /> | + | The '''Cas ENN''' proteins are part of '''CRISPR/Cas''' prokaryotic immune system which confers protection from foreign genetic elements like viruses. The '''CRISPR''' ('''C'''lustered '''R'''egularly '''I'''nterspersed '''S'''hort '''P'''alindromic '''R'''epeats) are DNA loci which are found in ca. 40% of the bacteria. The CRISPR/Cas system is being used lately as gene editing tool<ref>PMID:20056882</ref>. For more details see<br /> |
| | + | *[[CRISPR-Cas]] |
| | + | *CRISPR type I-A (Cascade) - see [[CRISPR subtype I-A]] |
| | + | *CRISPR type I-B (Cascade) - see [[CRISPR subtype I-B]] |
| | + | *CRISPR type I-C (Cascade) - see [[CRISPR subtype I-C]] |
| | + | *CRISPR type I-E (Cascade) - see [[CRISPR type I-E (Cascade)|CRISPR subtype I-E]] |
| | + | *(CRISPR type I-F (Csy1, Csy2, Csy3) - see [[CRISPR subtype I-F]] |
| | + | *CRISPR type III-A (Csm complex) - see [[CRISPR subtype III-A (Csm complex)]] |
| | + | *CRISPR type II-A - see [[CRISPR-Cas9]] |
| | + | *CRISPR type V (Cpf1, C2c1, C2c3) - see [[CRISPR type V]] |
| | + | *CRISPR type VI (Cas13a (previously known as C2c2), Cas13b, Cas13c, Cas13d) - see [[CRISPR type VI]] |
| | + | |
| | '''Intron-encoded ENN''' or '''homing ENN''' are encoded by genes with mobile, self-splicing introns. They promote the movement of DNA sequences from one chromosome location to another<ref>PMID:9665136</ref>. <br /> | | '''Intron-encoded ENN''' or '''homing ENN''' are encoded by genes with mobile, self-splicing introns. They promote the movement of DNA sequences from one chromosome location to another<ref>PMID:9665136</ref>. <br /> |
| | See also<br /> | | See also<br /> |
| Line 23: |
Line 34: |
| | | | |
| | <scene name='49/496318/Cv/2'>DNA-protein binding</scene> (PDB code [[1rva]]). | | <scene name='49/496318/Cv/2'>DNA-protein binding</scene> (PDB code [[1rva]]). |
| - | </StructureSection>
| + | |
| | ==3D structures of endonuclease== | | ==3D structures of endonuclease== |
| | + | [[Endonuclease 3D structures]] |
| | | | |
| - | Updated on {{REVISIONDAY2}}-{{MONTHNAME|{{REVISIONMONTH}}}}-{{REVISIONYEAR}}
| + | </StructureSection> |
| - | {{#tree:id=OrganizedByTopic|openlevels=0|
| + | |
| - | | + | |
| - | *ENN-I
| + | |
| - | | + | |
| - | **[[1bix]] - hENN – human<br />
| + | |
| - | **[[1cfr]] – ENN – ''Citrobacter freundii''<br />
| + | |
| - | **[[1af5]] - CrENN – ''Chlamydomonas reinhardtii''<br />
| + | |
| - | **[[1fzr]], [[1m0d]], [[1m0i]], [[3cae]] – T7ENN - Enterobacteria phage T7<br />
| + | |
| - | **[[2g7e]], [[2g7f]] – VcENN – ''Vibrio cholera''<br />
| + | |
| - | **[[2vnd]] - VcENN (mutant) <br />
| + | |
| - | **[[2pu3]] - VsENN – ''Vibrio salmonicida''
| + | |
| - | | + | |
| - | *ENN-II
| + | |
| - | | + | |
| - | **[[1pvi]], [[1pvu]] - ENN – ''Proteus vulgaris''<br />
| + | |
| - | **[[2wsh]] - T4ENN (mutant) - Enterobacteria phage T4<br />
| + | |
| - | **[[3w52]], [[4cwm]], [[4cxo]], [[4cxp]], [[4cxv]] – ENN – ''Arabidopsis thaliana''<br />
| + | |
| - | | + | |
| - | *ENN-III
| + | |
| - | | + | |
| - | **[[2abk]], [[1ako]] - EcENN – ''Escherichia coli''<br />
| + | |
| - | **[[1orn]], [[1orp]], [[1p59]] - GsENN + DNA – ''Geobacillus stearothermophilus''
| + | |
| - | | + | |
| - | *ENN-IV
| + | |
| - | | + | |
| - | **[[1qtw]] - EcENN <br />
| + | |
| - | **[[2nqh]] - EcENN (mutant) <br />
| + | |
| - | **[[1qum]] – EcENN + DNA<br />
| + | |
| - | **[[2nqj]], [[2nq9]], [[4k1g]] - EcENN (mutant) + DNA<br />
| + | |
| - | **[[1xp3]] - ENN – ''Bacillus anthracis''<br />
| + | |
| - | **[[3aal]] - GkENN – ''Geobacillus kaustophilus''<br />
| + | |
| - | **[[3aam]] - TtENN – ''Thermus thermophilus''
| + | |
| - | | + | |
| - | *ENN-V
| + | |
| - | | + | |
| - | **[[1eni]], [[1enj]], [[1enk]], [[2end]] - T4ENN <br />
| + | |
| - | **[[1vas]] - T4ENN + DNA<br />
| + | |
| - | **[[2w35]], [[2w36]], [[4b20]] - TmENN + DNA – ''Thermotoga maritima''<br />
| + | |
| - | **[[3hd0]] - TmENN <br />
| + | |
| - | **[[3ga2]] - BsENN (mutant) – ''Bacillus subtilis''<br />
| + | |
| - | **[[3goc]] - ENN – ''Streptomyces avermitilis''<br />
| + | |
| - | **[[2fcc]] – T4ENN + DNA<br />
| + | |
| - | **[[4nsp]] – hENN catalytic domain<br />
| + | |
| - | | + | |
| - | *ENN-VII
| + | |
| - | | + | |
| - | **[[1en7]], [[1e7d]] - T4ENN <br />
| + | |
| - | **[[1e7l]] - T4ENN (mutant) <br />
| + | |
| - | **[[2qnc]], [[2qnf]] - T4ENN (mutant) + DNA
| + | |
| - | | + | |
| - | *ENN-VIII
| + | |
| - | | + | |
| - | **[[1q39]], [[1q39]] - EcENN <br />
| + | |
| - | **[[1q3b]], [[1q3c]] - EcENN (mutant) <br />
| + | |
| - | **[[1tdh]] – hENN <br />
| + | |
| - | **[[1bix]] – hENN + DNA<br />
| + | |
| - | **[[1k3x]], [[2ea0]], [[1k3w]], [[1k3x]], [[2ea0]], [[2opf]], [[2oq4]] - EcENN + DNA <br />
| + | |
| - | **[[2opf]], [[2oq4]] - EcENN (mutant) + DNA
| + | |
| - | | + | |
| - | *AP ENN
| + | |
| - | | + | |
| - | **[[3ngf]] - ENN - ''Brucella melitensis''<br />
| + | |
| - | **[[2j63]] – ENN – ''Leishmania major''<br />
| + | |
| - | | + | |
| - | *Cas ENN
| + | |
| - | | + | |
| - | **[[1wj9]], [[4ej3]] – TtCas<br />
| + | |
| - | **[[4n06]] – AfCas1 - ''Archaeoglobus fulgidus'' <br />
| + | |
| - | **[[3god]] – PaCas1 – ''Pseudomonas aeruginosa''<br />
| + | |
| - | **[[3nkd]] – EcCas1<br />
| + | |
| - | **[[4wj0]] – PhCas1 - ''Pyrococcus horikoshii''<br />
| + | |
| - | **[[4xtk]] – TmCas1<br />
| + | |
| - | **[[3oq2]] – Cas2 – ''Desulfovibrio vulgaris''<br />
| + | |
| - | **[[4mak]] – EcCas2<br />
| + | |
| - | **[[4p6i]], [[4qdl]] – EcCas1 + EcCas2<br />
| + | |
| - | **[[3s4l]] – MjCas3 – ''Methanocaldococcus jannaschii''<br />
| + | |
| - | **[[4qqw]] – TfCas3 – ''Thermobifida fusca''<br />
| + | |
| - | **[[4qqx]] – TfCas3 + ATP <br />
| + | |
| - | **[[4qqy]] – TfCas3 + ADP <br />
| + | |
| - | **[[4qqz]] – TfCas3 + AMPPNP <br />
| + | |
| - | **[[4q2c]] - TteCas3 – ''Thermobaclum terrenum''<br />
| + | |
| - | **[[4q2d]] - TteCas3 + ATP<br />
| + | |
| - | **[[3i4h]] – Cas4 - ''Pyrobaculum calidifontis''<br />
| + | |
| - | **[[4qr0]], [[4qr1]] - SpCas9 (Csn1) – ''Streptococcus pyogenes''<br />
| + | |
| - | **[[4ill]], [[4ilm]] – SsCas6 + RNA - ''Sulfolobus solfataricus''<br />
| + | |
| - | **[[4ilr]], [[3zfv]] – SsCas6 <br />
| + | |
| - | **[[3i4h]] – PfCas6 - ''Pyrococcus furiosus''<br />
| + | |
| - | **[[3pkm]] – PfCas6 + RNA<br />
| + | |
| - | **[[4dzd]] – EcCas6e<br />
| + | |
| - | **[[2y8w]], [[2y8y]], [[3qrp]], [[3qrq]], [[2qrr]] – TtCas6e (Cse3) + RNA<br />
| + | |
| - | **[[2xli]], [[2xlj]], [[2xlk]], [[4al7]] – PaCas6f (Csy4) + RNA <br />
| + | |
| - | **[[4al5]], [[4al6]] - PaCas6f (Csy4) (mutant) + RNA <br />
| + | |
| - | **[[4cmp]], [[4cmq]] – SpCas9 (Csn1)<br />
| + | |
| - | **[[4oo8]], [[4un3]], [[4un4]], [[4un5]] - SpCas9 (Csn1) (mutant) + RNA + DNA<br />
| + | |
| - | **[[4ogc]], [[4oge]] – Cas9 II-C – Actinomyces naeslundii<br />
| + | |
| - | **[[3ung]], [[3ur3]], [[4doz]], [[4w8y]] – PfCas10 CMR2 subunit <br />
| + | |
| - | **[[4h4k]] – PfCas10 CMR2+CMR3 subunits <br />
| + | |
| - | **[[3w2v]] – PfCas10 CMR2+CMR3 subunits + AMP<br />
| + | |
| - | **[[3w2w]] – PfCas10 CMR2+CMR3 subunits + ATP<br />
| + | |
| - | **[[4gkf]] – PfCas10 CMR5 subunit<br />
| + | |
| - | **[[4an8]], [[4f3e]] – TtCasA (Cse1)<br />
| + | |
| - | **[[4h3t]] - CasA (Cse1) – ''Acidimicrobium ferrooxidans''<br />
| + | |
| - | **[[4h79]] - TfCasB (Cse2)<br />
| + | |
| - | **[[3wa8]] - CasB (Cse2) – ''Meiothermus ruber''<br />
| + | |
| - | **[[4h7a]] - TtCasB (Cse2)<br />
| + | |
| - | **[[4tvx]] – EcCasA (Cse1) + CasE<br />
| + | |
| - | **[[4u7u]] – EcCasA (Cse1) + CasB + CasC + CasD + CasE + RNA<br />
| + | |
| - | **[[4qyz]] – EcCasA (Cse1) + CasB + CasC + CasD + CasE + RNA + DNA<br />
| + | |
| - | **[[3qhq]] - Csn2 – ''Streptococcus agalactiae''<br />
| + | |
| - | **[[3toc]], [[3v7f]] – SpCsn2<br />
| + | |
| - | **[[3ps0]] – SsCsa2<br />
| + | |
| - | | + | |
| - | *EcoKI ENN
| + | |
| - | | + | |
| - | **[[2ar0]] – EcENN (mutant)<br />
| + | |
| - | | + | |
| - | *Ecoo109I ENN
| + | |
| - | | + | |
| - | **[[1wtd]] - EcENN<br />
| + | |
| - | **[[1wte]] – EcENN + DNA<br />
| + | |
| - | | + | |
| - | *EcoRI ENN
| + | |
| - | | + | |
| - | **[[1qc9]] - EcENN<br />
| + | |
| - | **[[1eri]], [[1ckq]], [[1cl8]], [[1qps]] – EcENN + DNA<br />
| + | |
| - | **[[1qrh]], [[1qri]], [[2oxv]] - EcENN (mutant) + DNA
| + | |
| - | | + | |
| - | *EcoRII ENN
| + | |
| - | | + | |
| - | **[[1na6]] – EcENN<br />
| + | |
| - | **[[3hqf]], [[3hqg]] - EnENN + DNA
| + | |
| - | | + | |
| - | *EcoRV ENN
| + | |
| - | | + | |
| - | **[[1rve]], [[1az3]] – EcENN<br />
| + | |
| - | **[[1az4]] - EcENN (mutant) <br />
| + | |
| - | **[[2rve]], [[4rve]], [[1rva]], [[1rvb]], [[1rvc]], [[1az0]], [[1bsu]], [[1bua]], [[1bgb]], [[1rv5]], [[1b94]], [[1b95]], [[1eo3]], [[1eo4]], [[1eon]], [[1eoo]], [[1eop]], [[2b0d]], [[2b0e]] - EcENN + DNA<br />
| + | |
| - | **[[1bss]], [[1b96]], [[1b97]], [[1stx]], [[1suz]], [[1sx5]], [[1sx8]], [[2ge5]] - EcENN (mutant) + DNA
| + | |
| - | | + | |
| - | *BamHI ENN
| + | |
| - | | + | |
| - | **[[1bam]] – BaENN – ''Bacillus amyloliquefaciens''<br />
| + | |
| - | **[[1bhm]], [[3bam]], [[2bam]], [[1esg]] - BaENN + DNA
| + | |
| - | | + | |
| - | *FokI ENN
| + | |
| - | | + | |
| - | **[[1fok]] – PoENN + DNA – ''Planomicrobium okeanokoites''<br />
| + | |
| - | **[[2fok]] – PoENN
| + | |
| - | | + | |
| - | *PvuII ENN
| + | |
| - | | + | |
| - | **[[3pvi]] - PvENN (mutant) + DNA – ''Proteus vulgaris''<br />
| + | |
| - | **[[1k0z]], [[1h56]], [[3ksk]] - PvENN<br />
| + | |
| - | **[[1ni0]] - PvENN (mutant) <br />
| + | |
| - | **[[2pvi]], [[1eyu]], [[1f0o]] - PvENN + DNA
| + | |
| - | | + | |
| - | *tRNA-splicing ENN
| + | |
| - | | + | |
| - | **[[1a79]], [[1a76]], [[1a77]] - MjENN – ''Methanocaldococcus jannaschii''<br />
| + | |
| - | **[[2cv8]] - ENN – ''Sulfolobus tokodaii''<br />
| + | |
| - | **[[2gw6]] – hENN subunit sen15 – NMR<br />
| + | |
| - | **[[2gjw]] - AfENN + RNA –'' Archaeoglobus fulgidus''<br />
| + | |
| - | **[[1r0v]], [[1r11]] – AfENN
| + | |
| - | **[[2ohc]], [[2ohe]] - TaENN – ''Thermoplasma acidophilum''<br />
| + | |
| - | **[[2zyz]] – ENN – ''Pyrobaculum aerophilum''<br />
| + | |
| - | **[[3iey]] - ENN + NEQ261 – ''Nanoarchaeum equitans''<br />
| + | |
| - | **[[3ajv]] - ApENN (mutant) – ''Aeropyrum pernix''<br />
| + | |
| - | **[[3p1y]] - ApENN /AfENN<br />
| + | |
| - | **[[3p1z]] - ApENN + protein
| + | |
| - | | + | |
| - | *VSR ENN
| + | |
| - | | + | |
| - | **[[1vsr]] – EcENN<br />
| + | |
| - | **[[1cw0]] - EcENN + DNA
| + | |
| - | | + | |
| - | *BglI ENN
| + | |
| - | | + | |
| - | **[[1dmu]], [[1d2i]] – BsENN + DNA <br />
| + | |
| - | **[[1dfm]] - BsENN (mutant) + DNA
| + | |
| - | | + | |
| - | *BglII ENN
| + | |
| - | | + | |
| - | **[[1es8]] - BsENN
| + | |
| - | | + | |
| - | *MunI ENN
| + | |
| - | | + | |
| - | **[[1d02]] - ENN (mutant) + DNA - ''Mycoplasma''
| + | |
| - | | + | |
| - | *Sm ENN
| + | |
| - | | + | |
| - | **[[1smn]], [[1qae]], [[1ql0]], [[1g8t]] – ENN – ''Serratia marcescens''
| + | |
| - | | + | |
| - | *NaeI ENN
| + | |
| - | | + | |
| - | **[[1ev7]] - LaENN – ''Lechevalieria aerocolonigenes''<br />
| + | |
| - | **[[1iaw]] - LaENN + DNA
| + | |
| - | | + | |
| - | *NgoMIV ENN
| + | |
| - | | + | |
| - | **[[1fiu]], [[4abt]] - ENN + DNA – ''Neisseria gonorrheae''
| + | |
| - | | + | |
| - | *NEI ENN
| + | |
| - | | + | |
| - | **[[1tdh]] - hENN
| + | |
| - | | + | |
| - | *BsoBI ENN
| + | |
| - | | + | |
| - | **[[1dc1]] - GsENN + DNA
| + | |
| - | | + | |
| - | *Bse634I ENN
| + | |
| - | | + | |
| - | **[[1knv]] – GsENN<br />
| + | |
| - | **[[3v1z]] – GsENN + DNA<br />
| + | |
| - | **[[3v20]], [[3v21]] - GsENN (mutant) + DNA
| + | |
| - | | + | |
| - | *HincII ENN
| + | |
| - | | + | |
| - | **[[1kc6]], [[1tw8]], [[1xhu]], [[1xhv]], [[1tx3]], [[2gie]] - HiENN + DNA – ''Haemophilus influenza''<br />
| + | |
| - | **[[2gig]], [[2gih]], [[2gii]], [[2gij]], [[3e3y]], [[3e40]], [[3e41]], [[3e42]], [[3e43]], [[3e44]], [[3e45]], [[3ebc]] - HiENN (mutant) + DNA<br />
| + | |
| - | **[[2aud]] – HiENN
| + | |
| - | | + | |
| - | *HinDIII ENN
| + | |
| - | | + | |
| - | **[[2e52]], [[3a4k]] - HiENN + DNA
| + | |
| - | | + | |
| - | *HinP1I ENN
| + | |
| - | | + | |
| - | **[[1ynm]] – HiENN<br />
| + | |
| - | **[[2fkc]], [[2fkh]], [[2fl3]], [[2flc]] - HiENN + DNA
| + | |
| - | | + | |
| - | *Flap ENN
| + | |
| - | | + | |
| - | **[[1mc8]] - PhENN (mutant) <br />
| + | |
| - | **[[3ory]]– ENN – ''Desulfurococcus amylolyticus''<br />
| + | |
| - | **[[3q8k]], [[3q8l]], [[3q8m]] - hENN + DNA<br />
| + | |
| - | **[[1rxv]], [[1rxw]] - AfENN + DNA<br />
| + | |
| - | **[[1ul1]] - hENN + PCNA <br />
| + | |
| - | **[[2izo]] - SsENN + PCNA – ''Sulfolobus solfataricus''<br />
| + | |
| - | **[[1a76]], [[1a77]] - MjENN <br />
| + | |
| - | **[[1b43]] – PfENN – ''Pyrococcus furiosus''<br />
| + | |
| - | **[[1rxz]] - AfENN + PCNA <br />
| + | |
| - | **[[4wa8]] – ENN – ''Methanopyrus kandleri''<br />
| + | |
| - | | + | |
| - | *DNA mismatch ENN
| + | |
| - | | + | |
| - | **[[1odg]] - EcENN + DNA<br />
| + | |
| - | **[[1h7u]] – hENN<br />
| + | |
| - | | + | |
| - | *Vvn ENN
| + | |
| - | | + | |
| - | **[[1ouo]] – VvENN – ''Vibrio vulnificus''<br />
| + | |
| - | **[[1oup]], [[2ivk]] - VvENN + DNA
| + | |
| - | | + | |
| - | *BfiI ENN
| + | |
| - | | + | |
| - | **[[2c1l]] – ENN – ''Bacillus firmus''
| + | |
| - | | + | |
| - | *SfiI ENN
| + | |
| - | | + | |
| - | **[[2ezv]], [[2f03]] – ENN + DNA – synthetic
| + | |
| - | | + | |
| - | *Ecl18kI ENN
| + | |
| - | | + | |
| - | **[[2fqz]], [[2gb7]] – ENN + DNA – ''Enterobacter cloacae''
| + | |
| - | | + | |
| - | *SdaI ENN
| + | |
| - | | + | |
| - | **[[2ixs]] – ENN – ''Streptomyces diastaticus''
| + | |
| - | | + | |
| - | *PabI ENN
| + | |
| - | | + | |
| - | **[[2dvy]] – ENN – ''Pyrococcus abyssi''
| + | |
| - | | + | |
| - | *PspGI ENN
| + | |
| - | | + | |
| - | **[[3bm3]] – ENN – ''Pyrococcus''
| + | |
| - | | + | |
| - | *NHN ENN
| + | |
| - | | + | |
| - | **[[2qgp]], [[4h6d]] – ENN – ''Geobacter metallireducens''
| + | |
| - | | + | |
| - | *UV damage ENN
| + | |
| - | | + | |
| - | **[[2j6v]], [[3bzg]], [[3c0s]] – TtENN <br />
| + | |
| - | **[[3bzj]], [[3c0l]], [[3c0q]] - TtENN (mutant)<br />
| + | |
| - | **[[3tc3]] - SaENN <br />
| + | |
| - | **[[4gle]] - SaENN + DNA<br />
| + | |
| - | | + | |
| - | *NotI ENN
| + | |
| - | | + | |
| - | **[[3bvq]] - NoENN – ''Nocardia otitidiscaviarum''<br />
| + | |
| - | **[[3c25]] - NoENN + DNA
| + | |
| - | | + | |
| - | *BpujI ENN
| + | |
| - | | + | |
| - | **[[2vla]], [[3s1s]] – ENN – ''Bacillus pumilus''
| + | |
| - | | + | |
| - | *Sau3AI ENN
| + | |
| - | | + | |
| - | **[[2reu]] – ENN C terminal – ''Staphylococcus aureus''
| + | |
| - | | + | |
| - | *HpyI ENN
| + | |
| - | | + | |
| - | **[[3fc3]], [[3gox]] – HpENN99 + DNA – ''Helicobacter pylori''<br />
| + | |
| - | **[[3or3]] – HpENN88 + DNA
| + | |
| - | | + | |
| - | *PacI ENN
| + | |
| - | | + | |
| - | **[[3ldy]], [[3m7k]] - PaENN + DNA – ''Psdeudomonas alcaligenes''
| + | |
| - | | + | |
| - | *Crossover junction ENN
| + | |
| - | | + | |
| - | **[[2kpf]] - ENN – mouse<br />
| + | |
| - | **[[2ziw]], [[2zix]], [[2ziu]] – hENN EME1 + Mus81<br />
| + | |
| - | **[[2ziv]] – hENN EME1 (mutant) + Mus81<br />
| + | |
| - | **[[4p0p]], [[4p0q]], [[4p0r]], [[4p0s]] – hENN EME1 + Mus81 + DNA<br />
| + | |
| - | | + | |
| - | *OkrAI ENN
| + | |
| - | | + | |
| - | **[[3odh]] - ENN + DNA – ''Oceanobacter kriegii''
| + | |
| - | | + | |
| - | *BspD6I ENN
| + | |
| - | | + | |
| - | **[[2ewf]] - ENN – ''Bacillus''
| + | |
| - | | + | |
| - | *ThaI ENN
| + | |
| - | | + | |
| - | **[[3ndh]] - TaENN + DNA
| + | |
| - | | + | |
| - | *Xpf ENN
| + | |
| - | | + | |
| - | **[[1z00]], [[2a1j]] - hENN C terminal + DNA excision repair protein<br />
| + | |
| - | **[[2aq0]] – hENN residues 823-905 – NMR<br />
| + | |
| - | **[[2kn7]] – hENN residues 823-905 + DNA – NMR<br />
| + | |
| - | **[[2bgw]] - ApENN + DNA <br />
| + | |
| - | **[[2bhn]] - ApENN <br />
| + | |
| - | | + | |
| - | *'''Intron-encoded endonuclease'''
| + | |
| - | | + | |
| - | *I-PpoI ENN
| + | |
| - | | + | |
| - | **[[1a74]], [[1ipp]], [[1a73]], [[1cz0]] - PpENN + DNA – ''Physarum polycephalum''<br />
| + | |
| - | **[[1cyq]], [[1evw]], [[2o6m]] - PpENN (mutant) + DNA<br />
| + | |
| - | **[[1evx]] - PpENN
| + | |
| - | | + | |
| - | *I-CreI ENN
| + | |
| - | | + | |
| - | **[[1bp7]], [[1g9y]], [[1g9z]], [[1n3e]], [[1n3f]], [[2vbj]], [[2vbl]], [[2vbn]], [[2vbo]], [[4aqu]], [[4aqx]] - CrENN + DNA <br />
| + | |
| - | **[[1u0c]], [[1u0d]], [[1t9i]], [[1t9j]], [[2i3p]], [[2i3q]], [[4aab]], [[4aad]], [[4aae]], [[4aaf]], [[4aag]] - CrENN (mutant) + DNA<br />
| + | |
| - | **[[2o7m]] - CrENN
| + | |
| - | | + | |
| - | *I-CeuI ENN
| + | |
| - | | + | |
| - | **[[2ex5]] - ENN (mutant) + DNA – ''Chlamydomonas eugametos''
| + | |
| - | | + | |
| - | *I-DmoI ENN
| + | |
| - | | + | |
| - | **[[1b24]], [[2vs7]], [[2vs8]], [[4un7]], [[4un8]], [[4un9]], [[4una]], [[4unb]], [[4unc]], [[4ut0]], [[4d6n]], [[4d6o]] - DmENN + DNA – ''Desulfurococcus mobilis''<br />
| + | |
| - | **[[1mow]] – DmENN/CrENN + DNA
| + | |
| - | | + | |
| - | *I-AniI ENN
| + | |
| - | | + | |
| - | **[[1p8k]], [[2qoj]], [[3uvf]] - EnENN + DNA – ''Emericella nidulans''<br />
| + | |
| - | **[[3eh8]] - EnENN (mutant) + DNA
| + | |
| - | | + | |
| - | *I-HmuI ENN
| + | |
| - | | + | |
| - | **[[1u3e]] - ENN + DNA – Bacillus phage spo1
| + | |
| - | | + | |
| - | *I-TevI ENN
| + | |
| - | | + | |
| - | **[[1i3j]], [[1t2t]] - T4ENN + DNA<br />
| + | |
| - | **[[1ln0]], [[1mk0]] – T4ENN (mutant)
| + | |
| - | | + | |
| - | *I-SceI ENN
| + | |
| - | | + | |
| - | **[[1dfa]], [[1ef0]] – yENN – yeast<br />
| + | |
| - | **[[1gpp]] – yENN (mutant) <br />
| + | |
| - | **[[1lws]], [[1lwt]], [[1r7m]], [[3c0w]], [[3c0x]], [[3ool]] - yENN + DNA<br />
| + | |
| - | **[[3oor]] - yENN (mutant) + DNA
| + | |
| - | | + | |
| - | *I-PkoII ENN
| + | |
| - | | + | |
| - | **[[2cw7]], [[2cw8]] – ENN – ''Thermococcus kodakarensis''
| + | |
| - | | + | |
| - | *I-MspI ENN
| + | |
| - | | + | |
| - | **[[1sa3]], [[1yfi]], [[2fld]] – ENN + DNA – ''Moraxella''
| + | |
| - | | + | |
| - | *I-PfuI ENN
| + | |
| - | | + | |
| - | **[[1dq3]] – PfENN
| + | |
| - | | + | |
| - | *I-Tsp061I ENN
| + | |
| - | | + | |
| - | **[[2dch]] – ENN – ''Thermoproteus''
| + | |
| - | | + | |
| - | *I-Vdi141I ENN
| + | |
| - | | + | |
| - | **[[3e54]] – ENN – ''Vulcanisaeta distribute''
| + | |
| - | | + | |
| - | *I-MtuI ENN
| + | |
| - | | + | |
| - | **[[3ifj]], [[3igd]] - MtENN (mutant) – ''Mycobacterium tuberculosis''<br />
| + | |
| - | **[[2l8l]] - MtENN
| + | |
| - | | + | |
| - | *I-MsoI ENN
| + | |
| - | | + | |
| - | **[[1m5x]], [[3fd2]] – MoENN + DNA – ''Monomastix''<br />
| + | |
| - | **[[3ko2]] – MoENN (mutant) + DNA
| + | |
| - | | + | |
| - | *I-GzeMII ENN
| + | |
| - | | + | |
| - | **[[4efj]] – ENN + DNA – ''Gibberella zeae''
| + | |
| - | | + | |
| - | *I-tRNA ENN
| + | |
| | | | |
| - | **[[4fz2]] – MoENN – ''Candidatus micrarchaeum''<br /> | |
| - | }} | |
| | == References == | | == References == |
| | <references/> | | <references/> |
| Line 451: |
Line 45: |
| | | | |
| | [[Category:Topic Page]] | | [[Category:Topic Page]] |
| | + | [[Category:Endonuclease]] |