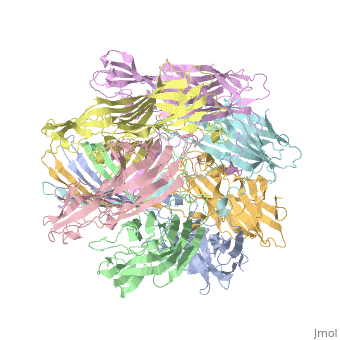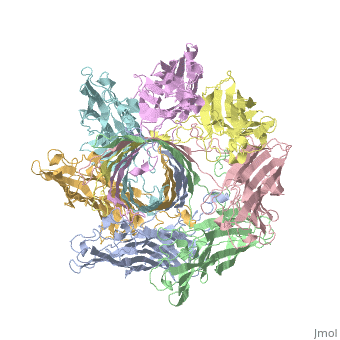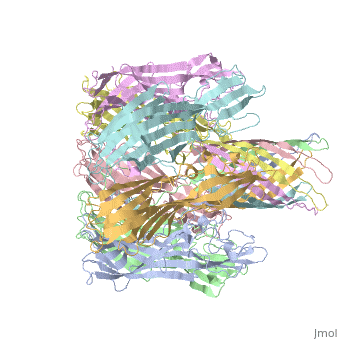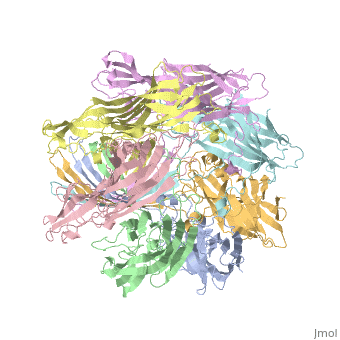This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Hemolysin
From Proteopedia
(Difference between revisions)
| (8 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
| - | + | <StructureSection load='7ahl' size='350' side='right' caption='α-hemolysin heptamer (PDB code [[7ahl]]).' scene=''> | |
== Function == | == Function == | ||
| - | '''Hemolysin''' (HL) is exotoxin from bacteria which causes lysis of red blood cells<ref>PMID:20110774</ref>. | + | '''Hemolysin''' (HL) is exotoxin from bacteria which causes lysis of red blood cells<ref>PMID:20110774</ref>. '''α-hemolysin''' from the bacterium ''Clostridium'' are called '''alpha-toxin'''. They are zinc metalloenzymes which bind to the membrane in the presence of calcium. It acts as a phospholipase C. |
See details for α-hemolysin in [[Pore forming toxin, α-hemolysin]]. | See details for α-hemolysin in [[Pore forming toxin, α-hemolysin]]. | ||
| Line 11: | Line 11: | ||
HL acts as a virulence factor in the pathogenesis of invasive infections<ref>PMID:12564994</ref>. | HL acts as a virulence factor in the pathogenesis of invasive infections<ref>PMID:12564994</ref>. | ||
| - | == 3D | + | ==3D Printed Physical Model of Hemolysin== |
| - | + | Shown below is a 3D printed physical model of Hemolysin. The model is shown in alpha carbon backbone format with each chain colored uniquely. | |
| - | + | ||
| - | + | [[Image:hemolysin1_centerForBioMolecularModeling.jpg|550px]] | |
| + | [[Image:hemolysin2_centerForBioMolecularModeling.jpg|550px]] | ||
| - | + | ====The MSOE Center for BioMolecular Modeling==== | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | [[Image:CbmUniversityLogo.jpg | left | 150px]] | |
| - | + | The [http://cbm.msoe.edu MSOE Center for BioMolecular Modeling] uses 3D printing technology to create physical models of protein and molecular structures, making the invisible molecular world more tangible and comprehensible. To view more protein structure models, visit our [http://cbm.msoe.edu/educationalmedia/modelgallery/ Model Gallery]. | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | == 3D Structures of hemolysin == | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | [[Hemolysin 3D structures]] | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | </StructureSection> | |
| - | **[[2wxt]], [[1ca1]] - CpAT + Cd + Zn – ''Clostridium perfringens''<br /> | ||
| - | **[[1qm6]], [[1gyg]], [[1kho]] - CpAT + Zn <br /> | ||
| - | **[[2wy6]], [[2wxu]] – CpAT (mutant) + Ca + Cd + Zn<br /> | ||
| - | **[[1qmd]] - CpAT + Ca + Zn<br /> | ||
| - | **[[1olp]] - AT + Ca + Zn – ''Clostridium absonum''<br /> | ||
| - | **[[2vk9]] - AT – ''Clostridium novyi''<br /> | ||
| - | }} | ||
== References == | == References == | ||
<references/> | <references/> | ||
[[Category:Topic Page]] | [[Category:Topic Page]] | ||
Current revision
| |||||||||||
References
- ↑ Mestre MB, Fader CM, Sola C, Colombo MI. Alpha-hemolysin is required for the activation of the autophagic pathway in Staphylococcus aureus-infected cells. Autophagy. 2010 Jan;6(1):110-25. PMID:20110774
- ↑ Nizet V. Streptococcal beta-hemolysins: genetics and role in disease pathogenesis. Trends Microbiol. 2002 Dec;10(12):575-80. PMID:12564994
Proteopedia Page Contributors and Editors (what is this?)
Michal Harel, Mark Hoelzer, Wayne Decatur, Alexander Berchansky