User:Brittany Stankavich/Sandbox 1
From Proteopedia
< User:Brittany Stankavich(Difference between revisions)
| (15 intermediate revisions not shown.) | |||
| Line 17: | Line 17: | ||
== Structure == | == Structure == | ||
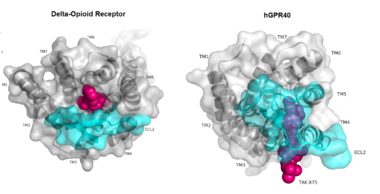
| - | hGPR40 is composed of seven-transmembrane helices that are characteristic of G-protein coupled receptors (GPCR)<ref name="crystal">PMID: 25043059</ref>. While there is relatively low sequence identity between hGPR40 and peptide-binding and opioid GPCRs, they do share structural similarities such as a conserved <scene name='72/727085/Hairpin_loop/ | + | hGPR40 is composed of seven-transmembrane helices that are characteristic of [https://en.wikipedia.org/wiki/G_protein%E2%80%93coupled_receptor G-protein coupled receptors] (GPCR)<ref name="crystal">PMID: 25043059</ref>. While there is relatively low sequence identity between hGPR40 and peptide-binding and [https://en.wikipedia.org/wiki/Opioid_receptor opioid GPCRs], they do share structural similarities such as a conserved <scene name='72/727085/Hairpin_loop/4'>hairpin loop</scene> motif on <scene name='72/727085/Ecl2/4'>extracellular loop 2 </scene>(ECL2)<ref name="crystal"/>. In addition, there is a conserved <scene name='72/727085/Disulfide/3'>disulphide bond</scene> that is formed between transmembrane helix 3 (Cys 79) and the C-terminus of ECL2 (Cys170)<ref name="crystal"/>. Compared to peptide-binding and opioid GPCRs which have distinctive [https://en.wikipedia.org/wiki/Beta_sheet β-sheets] spanning from transmembrane helix 4 to 5, hGPR40 possesses a shorter B-sheet-like region which has [http://proteopedia.org/wiki/index.php/Image:Beta-like_factors_of_hGPR40_ECL2.png low B-factors]<ref name="crystal"/>. This reflects the low mobility of the region that limits the overall flexibility of the adjacent portion of ECL2 between Leu171 and Asp175<ref name="crystal"/>. A unique feature of hGPR40 is the presence of an additional 13 residues (Pro147 to Gly159) on ECL2 which is absent on all the other peptide/opioid receptors<ref name="crystal"/>. These extra residues form a separate <scene name='72/727085/Auxiliary_loop/3'>auxiliary loop</scene> between the B-sheet-like region and transmembrane 4. Together, the auxiliary loop and ECL2 of hGPR40 function as a <scene name='72/727085/Ecl2_cap/3'>roof </scene> over the canonical binding site covering it from the central extracellular region<ref name="crystal"/>. |
| + | |||
| + | [[Image:Gpcr comparison.png|380 px|thumb|center|'''Figure 1:'''Comparison of Delta-opioid receptor to human free-fatty acid receptor (hGPR40) both of which are G-protein coupled receptors. The binding pocket of the delta-opioid receptor is solvent exposed allowing ligands to enter directly from the extracellular space while the binding pocket of hGPR40 is covered by the extracellular loop 2 (ECL2) preventing entry from the extracellular space (ECL2 represented in cyan). The Delta-opioid displays the canonical binding site typical of most GPCRs while ligands of hGPR40 bind to a noncanonical pocket represented in pink.]] | ||
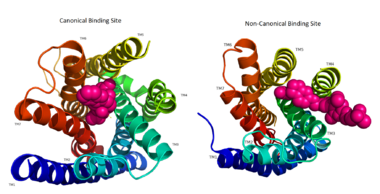
The canonical binding pocket for many other GPCRs is solvent exposed and centrally located between the transmembrane helices allowing ligands to directly bind from the extracellular space<ref name="crystal"/>. However, because the ECL2 acts as a roof to this canonical binding site, it inhibits ligands from entering directly from the extracellular region. Instead, the highly lipophilic nature of hGPRC40’s ligands allow it to enter a <scene name='72/727085/Hgpr40_entry/2'>noncanonical binding pocket </scene> between TM3 and TM4 by moving through the lipid bilayer<ref name="crystal"/>. | The canonical binding pocket for many other GPCRs is solvent exposed and centrally located between the transmembrane helices allowing ligands to directly bind from the extracellular space<ref name="crystal"/>. However, because the ECL2 acts as a roof to this canonical binding site, it inhibits ligands from entering directly from the extracellular region. Instead, the highly lipophilic nature of hGPRC40’s ligands allow it to enter a <scene name='72/727085/Hgpr40_entry/2'>noncanonical binding pocket </scene> between TM3 and TM4 by moving through the lipid bilayer<ref name="crystal"/>. | ||
| + | [[Image:Binding site comparison.png|380 px|thumb|center|'''Figure 2:''' Comparison of the canonical binding site represented in pink of most opioid/peptide binding GPCRs (left) compared to the noncanonical binding site of ligands with hGPR40 (right). ]] | ||
== Ligand Binding == | == Ligand Binding == | ||
| Line 27: | Line 30: | ||
GPR40’s natural substrate are FFAs in which a free [https://en.wikipedia.org/wiki/Carboxylic_acid carboxyl group] is required to bind. However, GPR40 can be activated by a wide variety of fatty acids with chain lengths ranging from [https://en.wikipedia.org/wiki/Saturated_fat saturated fatty acids] with 8 carbons to 23 carbons. In addition, various [https://en.wikipedia.org/wiki/Monounsaturated_fat mono] (i.e. [https://en.wikipedia.org/wiki/Palmitoleic_acid palmitoleic] (C16:1) and [https://en.wikipedia.org/wiki/Oleic_acid oleic] (C18:1) acids) and [https://en.wikipedia.org/wiki/Polyunsaturated_fatty_acid poly-unsaturated fatty acids] (i.e.[https://en.wikipedia.org/wiki/Linoleic_acid linoleic] (C18:2) and [https://en.wikipedia.org/wiki/List_of_unsaturated_fatty_acids#Eicosatrienoic_acid eicosatrienoic] (C20:3) acids) can activate GPR40 <ref name= "Morg"/>. The agonists potency varies according to the carbon-chain length however. The activity of GPR40 increases when the chain is increased from C6 to C15 but then decreased when the chain was extended beyond C15. One explanation for this is that as [https://en.wikipedia.org/wiki/Alkyl alkyl] chain increased, so did the [https://en.wikipedia.org/wiki/Hydrophobe hydrophobic] interactions with the protein within the binding pocket. However, for FFAs with carbon chains longer than C15, the molecular size is too large for the binding pocket. This causes the alkyl chain to extend beyond the binding pocket and destabilize the binding <ref name="Ren">PMID:26974599</ref>. | GPR40’s natural substrate are FFAs in which a free [https://en.wikipedia.org/wiki/Carboxylic_acid carboxyl group] is required to bind. However, GPR40 can be activated by a wide variety of fatty acids with chain lengths ranging from [https://en.wikipedia.org/wiki/Saturated_fat saturated fatty acids] with 8 carbons to 23 carbons. In addition, various [https://en.wikipedia.org/wiki/Monounsaturated_fat mono] (i.e. [https://en.wikipedia.org/wiki/Palmitoleic_acid palmitoleic] (C16:1) and [https://en.wikipedia.org/wiki/Oleic_acid oleic] (C18:1) acids) and [https://en.wikipedia.org/wiki/Polyunsaturated_fatty_acid poly-unsaturated fatty acids] (i.e.[https://en.wikipedia.org/wiki/Linoleic_acid linoleic] (C18:2) and [https://en.wikipedia.org/wiki/List_of_unsaturated_fatty_acids#Eicosatrienoic_acid eicosatrienoic] (C20:3) acids) can activate GPR40 <ref name= "Morg"/>. The agonists potency varies according to the carbon-chain length however. The activity of GPR40 increases when the chain is increased from C6 to C15 but then decreased when the chain was extended beyond C15. One explanation for this is that as [https://en.wikipedia.org/wiki/Alkyl alkyl] chain increased, so did the [https://en.wikipedia.org/wiki/Hydrophobe hydrophobic] interactions with the protein within the binding pocket. However, for FFAs with carbon chains longer than C15, the molecular size is too large for the binding pocket. This causes the alkyl chain to extend beyond the binding pocket and destabilize the binding <ref name="Ren">PMID:26974599</ref>. | ||
| - | FFAs bind to hGPR40 by coordinating its free carboxyl group to three amino acids, <scene name='72/727085/Ffa_binding/1'>Arg183, Tyr2240, and Arg258</scene>, which are located close to the <scene name='72/727085/Hgpr40_transmane_active/1'>extracellular domain</scene> of hGPR40 on TM5, 6 and 7. Because of the close proximity of these residues to the extracellular domain and the dominantly hydrophobic nature of FFA’s, it is | + | FFAs bind to hGPR40 by coordinating its free carboxyl group to three amino acids, <scene name='72/727085/Ffa_binding/1'>Arg183, Tyr2240, and Arg258</scene>, which are located close to the <scene name='72/727085/Hgpr40_transmane_active/1'>extracellular domain</scene> of hGPR40 on TM5, 6 and 7. Because of the close proximity of these residues to the extracellular domain and the dominantly hydrophobic nature of FFA’s, it is likely that ligand binding occurs close to the plane of the membrane <ref name="Morg">PMID:19660440</ref>. |
=== [http://proteopedia.org/wiki/index.php/Image:Tak_875.png TAK-875] === | === [http://proteopedia.org/wiki/index.php/Image:Tak_875.png TAK-875] === | ||
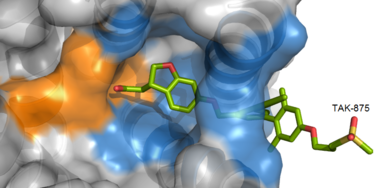
| - | <scene name='72/727085/Hgpr40_begin/3'> | + | <scene name='72/727085/Hgpr40_begin/3'>Tak-875</scene> is a [https://en.wikipedia.org/wiki/Partial_agonist partial agonist] of GPR40 and tested for the treatment of type 2 diabetes. The binding of TAK-875 to hGPR40 occurs by the ligand entering the binding site through the [https://en.wikipedia.org/wiki/Cell_membrane membrane bilayer]. This membrane insertion is performed via a method similar to ligand binding to [https://en.wikipedia.org/wiki/Sphingosine-1-phosphate_receptor sphingosine 1-phosphate receptor 1], retinal loading of [http://proteopedia.org/wiki/index.php/4j4q GPCR opsin], and the entry of anandamide in [https://en.wikipedia.org/wiki/Cannabinoid_receptor cannabinoid receptors], in which the <scene name='72/727085/Ecl2/4'>extracellular loops</scene> block the binding from the extracellular matrix <ref>PMID:22344443</ref>. The binding mechanism through the bilayer may be selectively favoring the free fatty acid because of the [https://en.wikipedia.org/wiki/Chemical_polarity#Nonpolar_molecules non-polar] regions of the ligand (Figure 2). |
| - | + | [[Image:Hydrophobic crop.png|380 px|center|thumb|'''Figure 3:''' TAK-875 buries its polar head group within a very hydrophobic region and coordinate within the polar charge network of hGPR40.]] | |
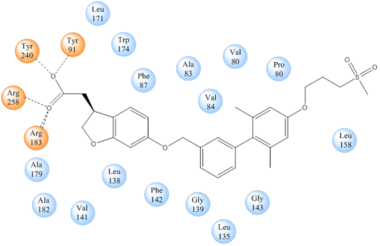
| - | TAK-875 binds to | + | TAK-875 binds to the <scene name='72/727085/Hgpr40_entry/2'>noncanonical binding site </scene> created between transmembrane (TM) domains 3-5 and the extracellular loop 2 (ECL2) of hGPR40. The ECL2 and auxiliary loop form a roof causing TAK-875 to enter through TM3 and TM4, first passing through the lipid bilayer. The carboxylate of TAK-875 is buried within a very hydrophobic region and in a complex complex <scene name='72/727085/Hgpr40_binding_relay/6'>charge network</scene> involving Glu172, Ser187, Asn241, and Asn 244 from hGPR40 forming ionic and polar interactions by coordinating TAK-875 with Arg183, Arg258, Tyr91, and Tyr240. |
| - | + | ||
| - | + | ||
| - | + | [[Image:Simplified hydrophibic.png|380 px|center|thumb|'''Figure 4:''' Stabilizing binding interactions between TAK-875 and hGPR40. Amino acids denoted in orange coordinate with the polar head of TAK-875 by polar/ionic interactions while blue amino acids stabilize the binding through hydrophobic interactions.]] | |
| - | [[Image:Simplified hydrophibic.png|380 px|center|thumb|Stabilizing binding interactions between TAK-875 and hGPR40. Amino acids denoted in orange coordinate with the polar head of TAK-875 by polar/ionic interactions while blue amino acids stabilize the binding through hydrophobic interactions.]] | + | |
== Signal Transduction == | == Signal Transduction == | ||
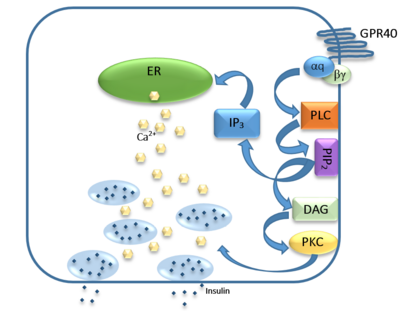
| - | [[Image:Gpr40 insulin pathway.png|400 px|center|thumb|Figure | + | [[Image:Gpr40 insulin pathway.png|400 px|center|thumb|'''Figure 5:''' Signaling pathway mediated by GPR40 in pancreatic β-cells. The 𝝰q subunit represents the Gq unit that is coupled to hGPR40. This subunit breaks off to activate phospholipase C (PLC), resulting in the hydrolysis of phosphatidylinositol-4,5-biphosphate (PIP<sub>2</sub>). PIP<sub>2</sub> goes on to create diacylglycerol (DAG) and inositol 1,4,5-triphosphate (IP3). The DAG activates protein kinase c (PKC), triggering secretion of insulin. Activated IP3 diffuses to the endoplasmic reticulum (ER), releasing the stored Ca<sup>2+</sup>, which aids in insulin secretion. ]] |
The natural substrate of hGPR40 is free fatty acids (FFAs) which bind to the G-protein and enhance glucose-stimulated insulin secretion <ref name="Morg">PMID:19660440</ref>. FFAs bind to GPR40 which then couples with the G-protein Gq leading to increased [https://en.wikipedia.org/wiki/Phospholipase_C phospholipase C] (PLC) activity<ref name="Morg"/>. PLC catalyzes the hydrolysis of the phospholipid [https://en.wikipedia.org/wiki/Phosphatidylinositol_4,5-bisphosphate phosphatidylinositol-4,5-biphosphate ] (PIP<sub>2</sub>) resulting in the formation of [https://en.wikipedia.org/wiki/Diglyceride diacylglycerol] (DAG) and [https://en.wikipedia.org/wiki/Inositol_trisphosphate inositol 1,4,5-triphosphate] (IP3)<ref name="Morg"/>. DAG can then activate [https://en.wikipedia.org/wiki/Protein_kinase_C protein kinase C] (PKC) to enhance insulin secretion<ref name="Morg"/>. IP3 on the other hand is soluble and diffuses to the [https://en.wikipedia.org/wiki/Endoplasmic_reticulum endoplasmic reticulum] where it binds a ligand-gated Ca<sup>2+</sup> channel<ref name="Morg"/>. This binding triggers the opening of the channel causing stored Ca<sup>2+</sup> to be released into the cytoplasm<ref name="Morg"/>. This large increase in intracellular free Ca<sup>2+</sup> produces a proportional increase in glucose-dependent insulin secretion, suggesting that insulin release can be contributed in part to the changes in Ca<sup>2+</sup> concentration resulting from activated GPR40 <ref name="Morg"/>. | The natural substrate of hGPR40 is free fatty acids (FFAs) which bind to the G-protein and enhance glucose-stimulated insulin secretion <ref name="Morg">PMID:19660440</ref>. FFAs bind to GPR40 which then couples with the G-protein Gq leading to increased [https://en.wikipedia.org/wiki/Phospholipase_C phospholipase C] (PLC) activity<ref name="Morg"/>. PLC catalyzes the hydrolysis of the phospholipid [https://en.wikipedia.org/wiki/Phosphatidylinositol_4,5-bisphosphate phosphatidylinositol-4,5-biphosphate ] (PIP<sub>2</sub>) resulting in the formation of [https://en.wikipedia.org/wiki/Diglyceride diacylglycerol] (DAG) and [https://en.wikipedia.org/wiki/Inositol_trisphosphate inositol 1,4,5-triphosphate] (IP3)<ref name="Morg"/>. DAG can then activate [https://en.wikipedia.org/wiki/Protein_kinase_C protein kinase C] (PKC) to enhance insulin secretion<ref name="Morg"/>. IP3 on the other hand is soluble and diffuses to the [https://en.wikipedia.org/wiki/Endoplasmic_reticulum endoplasmic reticulum] where it binds a ligand-gated Ca<sup>2+</sup> channel<ref name="Morg"/>. This binding triggers the opening of the channel causing stored Ca<sup>2+</sup> to be released into the cytoplasm<ref name="Morg"/>. This large increase in intracellular free Ca<sup>2+</sup> produces a proportional increase in glucose-dependent insulin secretion, suggesting that insulin release can be contributed in part to the changes in Ca<sup>2+</sup> concentration resulting from activated GPR40 <ref name="Morg"/>. | ||
| Line 48: | Line 48: | ||
== Medical Relevance == | == Medical Relevance == | ||
| - | While undergoing clinical trials, the use of TAK-875 in the treatment of diabetes mellitus was terminated in the III phase. This was due to observed liver toxicity | + | While undergoing clinical trials, the use of TAK-875 in the treatment of diabetes mellitus was terminated in the III phase. This was due to observed liver toxicity hypothesized to be connected to the effect of TAK-875 on [https://en.wikipedia.org/wiki/Bile_acid bile acids], achieved through the inhibition of the efflux of bile acids into bile<ref>PMID:26276582</ref>. Other molecules are currently in development for activating hGPR40 without adversely affecting liver function. One promising lead compound is 3-ethoxypropanoic acid, however this molecule must be modified as its [https://en.wikipedia.org/wiki/Half-life half-life] is very short due to the rapid [https://en.wikipedia.org/wiki/Redox oxidation] at the [https://en.wikipedia.org/wiki/Benzyl benzyl position] during metabolism <ref name="Tak">PMID:25815144</ref>. Additional research may hold the answer to effective treatment of type 2 diabetes and should be centered on the hGPR40 receptor because of its unique glucose dependent insulin secretion. |
Current revision
- User:Brittany Stankavich/Sandbox 1
hGPR40 Homo sapiens
| |||||||||||
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 Srivastava A, Yano J, Hirozane Y, Kefala G, Gruswitz F, Snell G, Lane W, Ivetac A, Aertgeerts K, Nguyen J, Jennings A, Okada K. High-resolution structure of the human GPR40 receptor bound to allosteric agonist TAK-875. Nature. 2014 Jul 20. doi: 10.1038/nature13494. PMID:25043059 doi:http://dx.doi.org/10.1038/nature13494
- ↑ Ren XM, Cao LY, Zhang J, Qin WP, Yang Y, Wan B, Guo LH. Investigation of the Binding Interaction of Fatty Acids with Human G Protein-Coupled Receptor 40 Using a Site-Specific Fluorescence Probe by Flow Cytometry. Biochemistry. 2016 Mar 17. PMID:26974599 doi:http://dx.doi.org/10.1021/acs.biochem.6b00079
- ↑ 3.0 3.1 3.2 3.3 3.4 Ichimura A, Hirasawa A, Hara T, Tsujimoto G. Free fatty acid receptors act as nutrient sensors to regulate energy homeostasis. Prostaglandins Other Lipid Mediat. 2009 Sep;89(3-4):82-8. doi:, 10.1016/j.prostaglandins.2009.05.003. Epub 2009 May 19. PMID:19460454 doi:http://dx.doi.org/10.1016/j.prostaglandins.2009.05.003
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 4.6 4.7 4.8 Morgan NG, Dhayal S. G-protein coupled receptors mediating long chain fatty acid signalling in the pancreatic beta-cell. Biochem Pharmacol. 2009 Dec 15;78(12):1419-27. doi: 10.1016/j.bcp.2009.07.020., Epub 2009 Aug 4. PMID:19660440 doi:http://dx.doi.org/10.1016/j.bcp.2009.07.020
- ↑ Ren XM, Cao LY, Zhang J, Qin WP, Yang Y, Wan B, Guo LH. Investigation of the Binding Interaction of Fatty Acids with Human G Protein-Coupled Receptor 40 Using a Site-Specific Fluorescence Probe by Flow Cytometry. Biochemistry. 2016 Mar 17. PMID:26974599 doi:http://dx.doi.org/10.1021/acs.biochem.6b00079
- ↑ Hanson MA, Roth CB, Jo E, Griffith MT, Scott FL, Reinhart G, Desale H, Clemons B, Cahalan SM, Schuerer SC, Sanna MG, Han GW, Kuhn P, Rosen H, Stevens RC. Crystal structure of a lipid G protein-coupled receptor. Science. 2012 Feb 17;335(6070):851-5. PMID:22344443 doi:10.1126/science.1215904
- ↑ Li X, Zhong K, Guo Z, Zhong D, Chen X. Fasiglifam (TAK-875) Inhibits Hepatobiliary Transporters: A Possible Factor Contributing to Fasiglifam-Induced Liver Injury. Drug Metab Dispos. 2015 Nov;43(11):1751-9. doi: 10.1124/dmd.115.064121. Epub 2015, Aug 14. PMID:26276582 doi:http://dx.doi.org/10.1124/dmd.115.064121
- ↑ Takano R, Yoshida M, Inoue M, Honda T, Nakashima R, Matsumoto K, Yano T, Ogata T, Watanabe N, Hirouchi M, Yoneyama T, Ito S, Toda N. Discovery of DS-1558: A Potent and Orally Bioavailable GPR40 Agonist. ACS Med Chem Lett. 2015 Jan 13;6(3):266-70. doi: 10.1021/ml500391n. eCollection, 2015 Mar 12. PMID:25815144 doi:http://dx.doi.org/10.1021/ml500391n