This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Saposin
From Proteopedia
(Difference between revisions)
| (6 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
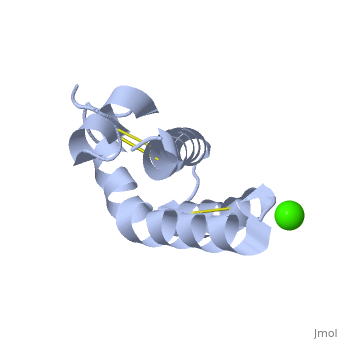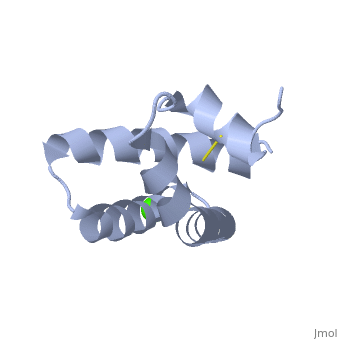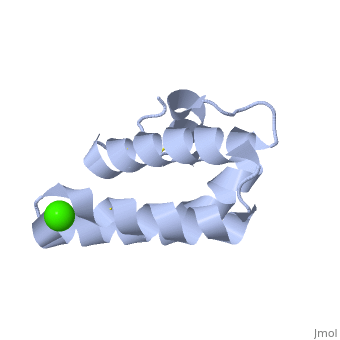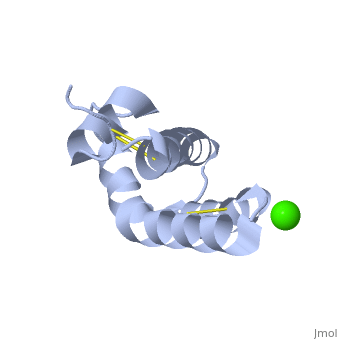
| - | + | <StructureSection load='2dob' size='350' side='right' scene='' caption='Human saposin A complex with Ca+2 [[2dob]]'> | |
== Function == | == Function == | ||
| - | '''Saposin''' (Sap) is a small protein which functions as activator of lipid-degrading enzymes. They act by isolating the lipid substrate from the membrane. Sap is synthesized as a precursor – prosaposin – which contain 4 SapB active domains (cleaved to saposin A,B,C and D) and 2 SapA domains which are cleaved off<ref>PMID:2001789</ref>. | + | '''Saposin''' (Sap) is a small protein which functions as activator of lipid-degrading enzymes. They act by isolating the lipid substrate from the membrane. Sap is synthesized as a precursor – prosaposin – which contain 4 SapB active domains (cleaved to saposin A,B,C and D) and 2 SapA domains which are cleaved off<ref>PMID:2001789</ref>. See also [[Lipid metabolism]]. |
*'''Saposin A and C''' stimulate hydrolysis of methylumbelliferyl β-galactoside by β-glucosylceramidase and of galactocerebrocide by β-galactosylceramidase<ref>PMID:2717620</ref>. For more details see [[Molecular Playground/Saposin C]].<br /> | *'''Saposin A and C''' stimulate hydrolysis of methylumbelliferyl β-galactoside by β-glucosylceramidase and of galactocerebrocide by β-galactosylceramidase<ref>PMID:2717620</ref>. For more details see [[Molecular Playground/Saposin C]].<br /> | ||
| Line 9: | Line 9: | ||
== Disease == | == Disease == | ||
Mutations in saposin B are autosomal recessive trait resulting in clinical metachromatic leukodystrophy<ref>PMID:17616409</ref>. Mutations in saposin D cause urinary system defects<ref>PMID:15345707</ref>. | Mutations in saposin B are autosomal recessive trait resulting in clinical metachromatic leukodystrophy<ref>PMID:17616409</ref>. Mutations in saposin D cause urinary system defects<ref>PMID:15345707</ref>. | ||
| - | + | </StructureSection> | |
== 3D Structures of Saposin == | == 3D Structures of Saposin == | ||
| Line 15: | Line 15: | ||
{{#tree:id=OrganizedByTopic|openlevels=0| | {{#tree:id=OrganizedByTopic|openlevels=0| | ||
| - | *Saposin A | + | *Saposin A residues 60-143 |
| - | **[[2dob]] – hSapA | + | **[[2dob]], [[4uex]] – hSapA – human<br /> |
**[[4ddj]] – hSapA + LDAO<br /> | **[[4ddj]] – hSapA + LDAO<br /> | ||
| + | **[[7p4d]] – mSapA – mouse<br /> | ||
| + | **[[5nxb]] – mSapA + galactocerebrosidase <br /> | ||
| + | |||
| + | *Saposin A residues 1-81 | ||
| + | |||
| + | **[[6d80]] – hSapA + mitochondrial calcium uniporter<br /> | ||
*Saposin B | *Saposin B | ||
| - | **[[1n69]] – hSapB + lipid | + | **[[1n69]] – hSapB + lipid<br /> |
| + | **[[4v2o]] – hSapB + chloroquine<br /> | ||
*Saposin C | *Saposin C | ||
| Line 32: | Line 39: | ||
**[[3bqp]], [[3bqq]], [[2r1q]], [[2rb3]] - hSapD residues 405-484<br /> | **[[3bqp]], [[3bqq]], [[2r1q]], [[2rb3]] - hSapD residues 405-484<br /> | ||
| + | **[[5u85]] - mSapD residues 438-519<br /> | ||
}} | }} | ||
== References == | == References == | ||
<references/> | <references/> | ||
[[Category:Topic Page]] | [[Category:Topic Page]] | ||
Current revision
| |||||||||||
3D Structures of Saposin
Updated on 14-November-2023
References
- ↑ O'Brien JS, Kishimoto Y. Saposin proteins: structure, function, and role in human lysosomal storage disorders. FASEB J. 1991 Mar 1;5(3):301-8. PMID:2001789
- ↑ Morimoto S, Martin BM, Yamamoto Y, Kretz KA, O'Brien JS, Kishimoto Y. Saposin A: second cerebrosidase activator protein. Proc Natl Acad Sci U S A. 1989 May;86(9):3389-93. PMID:2717620
- ↑ Yuan W, Qi X, Tsang P, Kang SJ, Illarionov PA, Besra GS, Gumperz J, Cresswell P. Saposin B is the dominant saposin that facilitates lipid binding to human CD1d molecules. Proc Natl Acad Sci U S A. 2007 Mar 27;104(13):5551-6. Epub 2007 Mar 19. PMID:17372201 doi:http://dx.doi.org/10.1073/pnas.0700617104
- ↑ Azuma N, O'Brien JS, Moser HW, Kishimoto Y. Stimulation of acid ceramidase activity by saposin D. Arch Biochem Biophys. 1994 Jun;311(2):354-7. PMID:8203897
- ↑ Deconinck N, Messaaoui A, Ziereisen F, Kadhim H, Sznajer Y, Pelc K, Nassogne MC, Vanier MT, Dan B. Metachromatic leukodystrophy without arylsulfatase A deficiency: a new case of saposin-B deficiency. Eur J Paediatr Neurol. 2008 Jan;12(1):46-50. Epub 2007 Jul 5. PMID:17616409 doi:http://dx.doi.org/10.1016/j.ejpn.2007.05.004
- ↑ Matsuda J, Kido M, Tadano-Aritomi K, Ishizuka I, Tominaga K, Toida K, Takeda E, Suzuki K, Kuroda Y. Mutation in saposin D domain of sphingolipid activator protein gene causes urinary system defects and cerebellar Purkinje cell degeneration with accumulation of hydroxy fatty acid-containing ceramide in mouse. Hum Mol Genet. 2004 Nov 1;13(21):2709-23. Epub 2004 Sep 2. PMID:15345707 doi:http://dx.doi.org/10.1093/hmg/ddh281