We apologize for Proteopedia being slow to respond. For the past two years, a new implementation of Proteopedia has been being built. Soon, it will replace this 18-year old system. All existing content will be moved to the new system at a date that will be announced here.
Zepatier
From Proteopedia
(Difference between revisions)
| (51 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
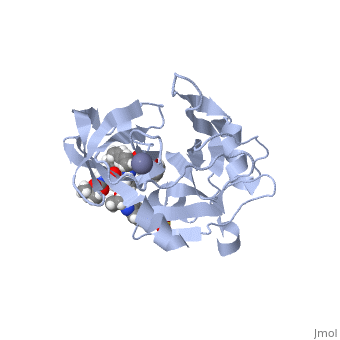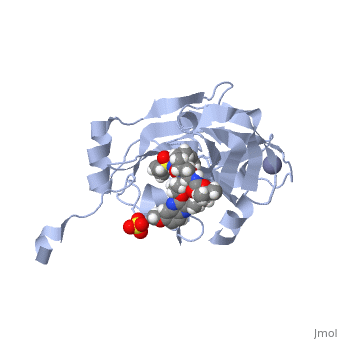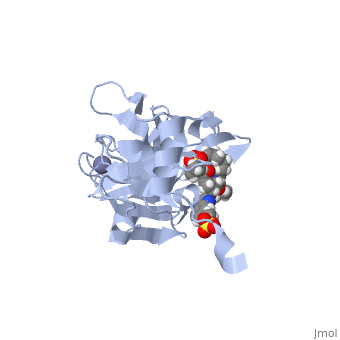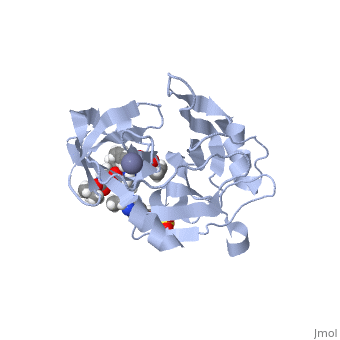
| - | + | <StructureSection load='5EPY' size='340' side='right' caption='Zepatier: A combination drug composed of Elbasvir and Grazoprevir.' scene=''> | |
| + | Zepatier [https://www.zepatier.com/?&gclid=CjwKEAiAmo_CBRC9qbGQssjqi28SJABYTgZxN1G3M7zIrEs1Escw2ai11BHclZaibEnrVK3yyEmrhhoCgUnw_wcB] is a combination drug composed of 50 mg elbasvir and 100 mg grazoprevir, combating genotypes 1 and 4 of chronic hepatitis C viral (HCV) infections in adults. | ||
| + | |||
| + | == Disease in Humans == | ||
| + | |||
| + | Hepatitis C [http://www.cdc.gov/hepatitis/hcv/index.htm] is a viral infection that causes inflammation of the liver, decreasing liver functioning. Decrease in liver function can in turn cause renal dysfunction. First isolated by Michael Houghton in 1989, hepatitis C was discovered to encode a single polyprotein, a large protein that is later cleaved into smaller proteins. The resulting polyprotein contains 3,000 amino acids, later cleaved into 10 polypeptides. Polypeptides produced include p7 ion channel proteins, the core protein, glycoproteins envelope 1 (E1) and envelope 2 (E2), and nonstructural proteins NS1, NS2, NS3, NS4A, NS5A, and NS5B. The 5’ untranslated region of HCV is not capped, allowing it to fold into a complex secondary RNA structure hereby creating an internal ribosome entry site that is able to direct ribosomal subunits and cellular factors, and as a result, translation<ref>Tan, S.-L. Hepatitis C viruses: genomes and molecular biology; Horizon bioscience: Wymondham, 2006.</ref>. Development of cell culture based model systems have allowed for the study of the HCV lifecycle <ref>Chevaliez, S.; Pawlotsky, J. M. Virology of hepatitis C virus infection. Best Pract. Res. Clin. Gastroenterol.2012, 26, 381-389.</ref>. | ||
| + | |||
| + | HCV is transmitted exclusively through blood whereupon the virus travels to the hepatocytes of the liver to undergo rapid replication<ref>Tan, S.-L. Hepatitis C viruses: genomes and molecular biology; Horizon bioscience: Wymondham, 2006.</ref>. This results in the inflammation of the liver, leading to decreased function and/or liver failure. HCV induces an immune response within the infected individual which commonly fails to restrict the development of chronicity. Six distinct genotype of HCV were discovered, genotype 1 is the most prevalent amongst populations and the most difficult to treat. Increased resistance of the virus at the M28, Q30, L31, or Y93 amino acids results in decreased effectiveness of pharmaceuticals against genotype 1. Genotype 1 results from the infection of human lymphoid cells by the virus. Genotype 4 has recently increased in prevalence, resulting in attempts to decrease transmission<ref>Tan, S.-L. Hepatitis C viruses: genomes and molecular biology; Horizon bioscience: Wymondham, 2006.</ref>. | ||
== Function == | == Function == | ||
| - | Zepatier is a combination drug combating genotypes 1 and 4 of chronic hepatitis C viral (HCV) infections in adults. Elbasvir inhibits NS5A, a non-enzymatic protein essential for viral replication and assembly of hepatitis C whose exact mechanism of action is unknown. Elbasvir | + | Zepatier is a combination drug combating genotypes 1 and 4 of chronic hepatitis C viral (HCV) infections in adults. It is designed for the treatment of individuals with end stage renal disease on dialysis, typically genotype 1 infected patients. The drug is also utilized to treat genotype 4 patients when other methods of treatment prove ineffective <ref>Petrescu, I. O.; Biciusca, V.; Taisescu, C. I.; Alexandru, D. O.; Taisescu, O.; Comanescu, M. V.; Petrescu, F.; Popescu, I. A.; Trasca, D. M.; ForTofoiu, M. C.; Silosi, C. A.; ForTofoiu, M. Histological factors that predict the liver fibrosis in patients with chronic hepatitis C. Rom. J. Morphol. Embryol. 2016, 57, 759-765.</ref>. For an example of the HCV genome, click here [https://www.ncbi.nlm.nih.gov/books/NBK1630/figure/ch1.f2/?report=objectonly]. <scene name='74/746120/Elbasvir_structure3/1'>Elbasvir</scene> inhibits NS5A, a non-enzymatic protein essential for viral replication and assembly of hepatitis C whose exact mechanism of action is unknown. Elbasvir restricts viral production at the early stage of assembly, in turn preventing spread of the virus<ref name="elbasvir">DOI: 10.1002/cmdc.201300343</ref>. |
| - | Grazoprevir is a macrocyclic compound that is able to reversibly bind to the protease, NS3/4A | + | <scene name='74/746120/Grazoprevir_meg/1'>Grazoprevir</scene> is a macrocyclic compound that is able to reversibly bind to the protease, NS3/4A, essential for viral replication. It is responsible for cleavage and processing of the HCV polyprotein. It is hypothesized that the protease is utilized to circumvent immune response at the primary stages of infection <ref>DOI: 10.1021/acschembio.5b00647</ref>. |
== Structure and Mechanism of Elbasvir == | == Structure and Mechanism of Elbasvir == | ||
| - | <scene name='74/745998/Elbasvir_structure1/7'>Elbasvir</scene>, MK-8742,a tetra-cyclic indole based NS5A inhibitor, was experimentally obtained through the modification of MK-4882. MK-4882 was one of the first clinical candidate drugs as an NS5A inhibitor. Although initially effective, viral breakthrough was a concern which led to | + | <scene name='74/745998/Elbasvir_structure1/7'>Elbasvir</scene>, MK-8742,a tetra-cyclic <scene name='74/746120/Indole_elbasvir/3'>indole</scene> based NS5A inhibitor, was experimentally obtained through the modification of MK-4882. MK-4882 was one of the first clinical candidate drugs as an NS5A inhibitor. Although initially effective, viral breakthrough was a concern which led to continued development of compounds effective against multiple genotypes as well as NS5A mutations. Through efficacy studies of infected chimpanzees, Elbasvir was discovered as a diastereomer of the known compound, MK-4882 <ref name="elbasvir" />. Elbasvir has a structural formula of C<sub>49</sub>H<sub>55</sub>N<sub>9</sub>O<sub>7</sub> and a molecular weight of 882.035 g/mol. It is a highly flexible protein with 13 rotatable bonds. It has four hydrogen bond donors and nine hydrogen bond acceptors <ref>National Center for Biotechnology Inforamtion. PubChem Compound Database; CID=71661251, https://pubchem.ncbi.nlm.nih.gov/compound/71661251#section=Chemical-and-Physical-Properties</ref>. |
| - | Although the actual mechanism of Elbasvir as a NS5A inhibitor is unknown, several hypothetical mechanisms have been proposed. The NS5A protein is | + | Although the actual mechanism of Elbasvir as a NS5A inhibitor is unknown, several hypothetical mechanisms have been proposed. The NS5A protein is critical for both DNA replication and assembly in the hepatitis C virus. With critical roles in both processes, NS5A is an excellent source as a target for inhibitors. NS5A is located within the endoplasmic reticulum of the cell. When inhibited, the protein is redistributed from the ER to lipid droplets. If the cellular location changed, NS5A would be unable to aid in viral replication, illustrating a form of protein inhibition caused by Elbasvir. Another potential mechanism of NS5A inhibitors is the altering of the phosphorylation of the NS5A protein <ref>http://dx.doi.org/10.1016/j.jhep.2013.03.030 |
| + | </ref>. The function of the NS5A protein is highly dependent on basal and hyperphosphorylation <ref>doi: 10.1128/JVI.00253-11</ref>. Since the phosphorylation of the NS5A protein is critical for protein function, altering levels of phosphorylation could impact the activity of the protein. | ||
== Structure and Mechanism of Grazoprevir == | == Structure and Mechanism of Grazoprevir == | ||
| - | <scene name='74/745998/Grazoprevir/3'>Grazoprevir</scene> | + | <scene name='74/745998/Grazoprevir/3'>Grazoprevir</scene> (C<sub>38</sub>H<sub>50</sub>N<sub>6</sub>O<sub>9</sub>, molecular weight: 766.911 g/mol) <ref>National Center for Biotechnology Information. PubChem Compound Database; CID=44603531, https://pubchem.ncbi.nlm.nih.gov/compound/44603531.</ref> stops the hepatitis C virus by inhibiting the NS3/4A protease, which is an enzyme responsible for the cleavage of the viral polyprotein. Cleavage of this polyprotein facilitates viral assembly and maturation of the virus. The NS3/4A protease is a protein complex in which the active site consists of a <scene name='74/746120/Catalytic_triad_m/1'>catalytic triad</scene> of amino acids S139, H57 and D81, and an activating cofactor which assists in, and is required for the activity of the protease. For more interactive models of the Grazoprevir, click here [http://www.rcsb.org/pdb/explore/explore.do?structureId=5EPY]. Grazoprevir has been proven to be more effective than similar protease inhibitors due to its two-way approach to inhibition. NS3/4A protease cleaves both the viral polyprotein of the Hep C virus and host factors involved in the immune response. Grazoprevir is unique in regard to its P4 cap, and <scene name='74/746120/P2-p4_macrocylic_ring/1'>P2 to P4 macrocyclic restriction</scene>, which interacts with the catalytic triad of the NS3/4A protease in its own distinct conformation. Due to this conformation, Grazoprevir can easily bind to the catalytic triad because it is already in a highly favorable arrangement, stopping the triads activity.<ref>DOI: 10.1021/acschembio.5b00647</ref>. |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| Line 33: | Line 30: | ||
== References == | == References == | ||
<references/> | <references/> | ||
| - | <ref>DOI: 10.1002/cmdc.201300343</ref> | + | <ref name="elbasvir">DOI: 10.1002/cmdc.201300343</ref> |
<ref>National Center for Biotechnology Inforamtion. PubChem Compound Database; CID=71661251, https://pubchem.ncbi.nlm.nih.gov/compound/71661251#section=Chemical-and-Physical-Properties</ref> | <ref>National Center for Biotechnology Inforamtion. PubChem Compound Database; CID=71661251, https://pubchem.ncbi.nlm.nih.gov/compound/71661251#section=Chemical-and-Physical-Properties</ref> | ||
| - | <ref>DOI: http://dx.doi.org/10.1016/j.jhep.2013.03.030</ref> | ||
<ref>doi: 10.1128/JVI.00253-11</ref> | <ref>doi: 10.1128/JVI.00253-11</ref> | ||
<ref>Chevaliez, S.; Pawlotsky, J. M. Virology of hepatitis C virus infection. Best Pract. Res. Clin. Gastroenterol.2012, 26, 381-389.</ref> | <ref>Chevaliez, S.; Pawlotsky, J. M. Virology of hepatitis C virus infection. Best Pract. Res. Clin. Gastroenterol.2012, 26, 381-389.</ref> | ||
| Line 41: | Line 37: | ||
<ref>DOI: 10.1021/acschembio.5b00647</ref> | <ref>DOI: 10.1021/acschembio.5b00647</ref> | ||
<ref>National Center for Biotechnology Information. PubChem Compound Database; CID=44603531, https://pubchem.ncbi.nlm.nih.gov/compound/44603531.</ref> | <ref>National Center for Biotechnology Information. PubChem Compound Database; CID=44603531, https://pubchem.ncbi.nlm.nih.gov/compound/44603531.</ref> | ||
| + | <ref>DOI: 10.1021/acschembio.5b00647</ref> | ||
| + | <ref>Tan, S.-L. Hepatitis C viruses: genomes and molecular biology; Horizon bioscience: Wymondham, 2006. </ref> | ||
| + | <ref>http://dx.doi.org/10.1016/j.jhep.2013.03.030</ref> | ||
Current revision
| |||||||||||
References
- ↑ Tan, S.-L. Hepatitis C viruses: genomes and molecular biology; Horizon bioscience: Wymondham, 2006.
- ↑ Chevaliez, S.; Pawlotsky, J. M. Virology of hepatitis C virus infection. Best Pract. Res. Clin. Gastroenterol.2012, 26, 381-389.
- ↑ Tan, S.-L. Hepatitis C viruses: genomes and molecular biology; Horizon bioscience: Wymondham, 2006.
- ↑ Tan, S.-L. Hepatitis C viruses: genomes and molecular biology; Horizon bioscience: Wymondham, 2006.
- ↑ Petrescu, I. O.; Biciusca, V.; Taisescu, C. I.; Alexandru, D. O.; Taisescu, O.; Comanescu, M. V.; Petrescu, F.; Popescu, I. A.; Trasca, D. M.; ForTofoiu, M. C.; Silosi, C. A.; ForTofoiu, M. Histological factors that predict the liver fibrosis in patients with chronic hepatitis C. Rom. J. Morphol. Embryol. 2016, 57, 759-765.
- ↑ 6.0 6.1 Coburn CA, Meinke PT, Chang W, Fandozzi CM, Graham DJ, Hu B, Huang Q, Kargman S, Kozlowski J, Liu R, McCauley JA, Nomeir AA, Soll RM, Vacca JP, Wang D, Wu H, Zhong B, Olsen DB, Ludmerer SW. Discovery of MK-8742: an HCV NS5A inhibitor with broad genotype activity. ChemMedChem. 2013 Dec;8(12):1930-40. doi: 10.1002/cmdc.201300343. Epub 2013 Oct, 14. PMID:24127258 doi:http://dx.doi.org/10.1002/cmdc.201300343
- ↑ Soumana DI, Kurt Yilmaz N, Prachanronarong KL, Aydin C, Ali A, Schiffer CA. Structural and Thermodynamic Effects of Macrocyclization in HCV NS3/4A Inhibitor MK-5172. ACS Chem Biol. 2015 Dec 18. PMID:26682473 doi:http://dx.doi.org/10.1021/acschembio.5b00647
- ↑ National Center for Biotechnology Inforamtion. PubChem Compound Database; CID=71661251, https://pubchem.ncbi.nlm.nih.gov/compound/71661251#section=Chemical-and-Physical-Properties
- ↑ http://dx.doi.org/10.1016/j.jhep.2013.03.030
- ↑ Fridell RA, Qiu D, Valera L, Wang C, Rose RE, Gao M. Distinct functions of NS5A in hepatitis C virus RNA replication uncovered by studies with the NS5A inhibitor BMS-790052. J Virol. 2011 Jul;85(14):7312-20. doi: 10.1128/JVI.00253-11. Epub 2011 May 18. PMID:21593143 doi:http://dx.doi.org/10.1128/JVI.00253-11
- ↑ National Center for Biotechnology Information. PubChem Compound Database; CID=44603531, https://pubchem.ncbi.nlm.nih.gov/compound/44603531.
- ↑ Soumana DI, Kurt Yilmaz N, Prachanronarong KL, Aydin C, Ali A, Schiffer CA. Structural and Thermodynamic Effects of Macrocyclization in HCV NS3/4A Inhibitor MK-5172. ACS Chem Biol. 2015 Dec 18. PMID:26682473 doi:http://dx.doi.org/10.1021/acschembio.5b00647
[1] [2] [3] [4] [5] [6] [7] [8] [9] [10]