This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Plasminogen activator
From Proteopedia
(Difference between revisions)
| (4 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
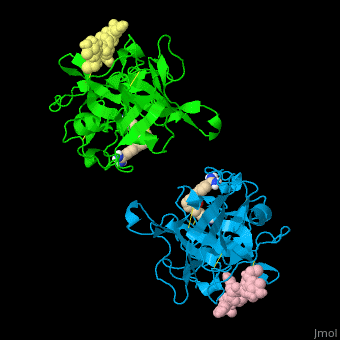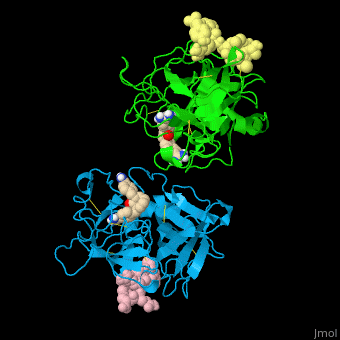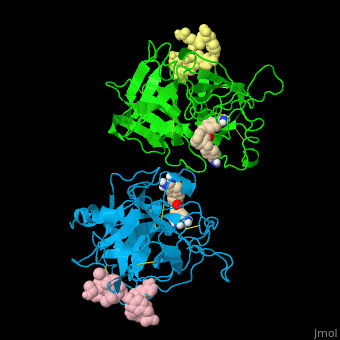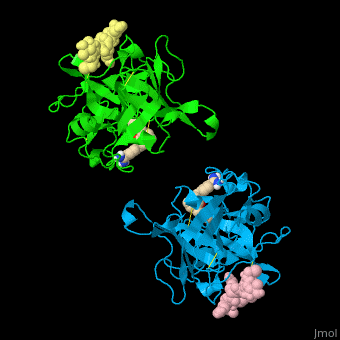
| - | <StructureSection load='' size=' | + | <StructureSection load='' size='350' side='right' caption='Human tissue plasminogen activator light chain catalytic domain (sky blue and green), heavy chain catalytic domain fragment (yellow and pink) complex with benzamidine, [[1a5h]]' scene='47/476004/Cv/1' pspeed='8' > |
== Function == | == Function == | ||
'''Plasminogen activator''' (PLA) is a serine protease which converts plasminogen to plasmin. TPA structure contains heavy and light chains (HC, LC). PLA is inhibited by PLA inhibitor 1 and 2. | '''Plasminogen activator''' (PLA) is a serine protease which converts plasminogen to plasmin. TPA structure contains heavy and light chains (HC, LC). PLA is inhibited by PLA inhibitor 1 and 2. | ||
| - | *'''Tissue PLA''' (TPA) which is involved in breakdown of blood clots | + | *'''Tissue PLA''' (TPA) which is involved in breakdown of blood clots. |
| + | *'''Urokinase-type PLA''' see [[Urokinase]].<br /> | ||
*'''Coagulase/fibrinolysin''' is a plasminogen activator from ''Yersinia pestis'' which possesses coagulase activity is responsible for degradation of plasmid-coded outer membrane proteins<ref>PMID:2843471</ref>. | *'''Coagulase/fibrinolysin''' is a plasminogen activator from ''Yersinia pestis'' which possesses coagulase activity is responsible for degradation of plasmid-coded outer membrane proteins<ref>PMID:2843471</ref>. | ||
| Line 9: | Line 10: | ||
Treatment with tissue PLA for acute ischemic stroke is beneficial<ref>PMID:7477192</ref>. | Treatment with tissue PLA for acute ischemic stroke is beneficial<ref>PMID:7477192</ref>. | ||
== Structural highlights == | == Structural highlights == | ||
| - | *<scene name='47/476004/Cv/ | + | *<scene name='47/476004/Cv/4'>Interactions between tissue plasminogen activator light chain catalytic domain and heavy chain catalytic domain fragment</scene>. Water molecules are shown as red spheres. |
| - | *<scene name='47/476004/Cv/ | + | *<scene name='47/476004/Cv/5'>Benzamidine binding site</scene>. |
</StructureSection> | </StructureSection> | ||
==3D structures of plasminogen activator== | ==3D structures of plasminogen activator== | ||
| Line 22: | Line 23: | ||
**[[1bqy]] – PLA – Pit viper | **[[1bqy]] – PLA – Pit viper | ||
| - | *Tissue plasminogen activator | + | *Tissue plasminogen activator; Domains – fibrin-binding 36-85; kringle 1 69-153; kringle 2 209-298; catalytic 311-561 |
**[[1a5h]] – hTPA HC+LC + benzamidine – human<br /> | **[[1a5h]] – hTPA HC+LC + benzamidine – human<br /> | ||
**[[1rtf]] - hTPA HC+LC<br /> | **[[1rtf]] - hTPA HC+LC<br /> | ||
**[[1tpg]] – hTPA F1-G - NMR<br /> | **[[1tpg]] – hTPA F1-G - NMR<br /> | ||
| - | **[[1bda]] – hTPA catalytic domain (mutant) + EGR-CMK<br /> | ||
**[[1pk2]] – hTPA kringle 2 domain + 6-aminohexanoic acid – NMR<br /> | **[[1pk2]] – hTPA kringle 2 domain + 6-aminohexanoic acid – NMR<br /> | ||
**[[1tpk]], [[1pml]] - hTPA kringle 2 domain<br /> | **[[1tpk]], [[1pml]] - hTPA kringle 2 domain<br /> | ||
| - | **[[1kdu]] - hTPA kringle domain – NMR<br /> | + | **[[1kdu]] - hTPA kringle 1 domain – NMR<br /> |
| - | **[[1tpm]], [[1tpn]] - hTPA fibrin-binding domain – NMR | + | **[[1tpm]], [[1tpn]] - hTPA fibrin-binding domain – NMR<br /> |
| + | **[[1bda]] – hTPA catalytic domain (mutant) + EGR-CMK<br /> | ||
| + | **[[5brr]], [[5zlz]] - hTPA catalytic domain (mutant) + plasminogen activator inhibitor 1<br /> | ||
| + | |||
| + | *Urokinase-type plasminogen activator | ||
| + | |||
| + | **[[4xsk]] - hUPA catalytic domain (mutant) <br /> | ||
| + | **[[5xg4]] - hUPA + quercetin<br /> | ||
| + | **[[5lhs]] - mUPA catalytic domain - mouse<br /> | ||
| + | **[[5lhn]], [[5lhp]], [[5lhq]], [[5lhr]], [[5hgg]] - mUPA catalytic domain (mutant) + antibody<br /> | ||
| + | **[[4zks]], [[4zkn]], [[4zko]], [[4zkr]] - hUPA catalytic domain (mutant) + UPAIN-1-W3A<br /> | ||
| + | **[[4zhl]], [[4x1s]], [[4x1r]], [[4x1q]], [[4x1p]], [[4x1n]], [[4x0w]] - hUPA catalytic domain (mutant) + MUPAIN-1-IG<br /> | ||
| + | **[[4zhm]] - hUPA catalytic domain (mutant) + MUPAIN-1-16-IG<br /> | ||
| + | **[[4os7]], [[4os6]], [[4os5]], [[4os4]], [[4os2]], [[4os1]], [[4mny]], [[4mnx]], [[4mnw]], [[4mnv]] - hUPA catalytic domain (mutant) + bicyclic peptide<br /> | ||
*Coagulase/fibrinolysin | *Coagulase/fibrinolysin | ||
Current revision
| |||||||||||
3D structures of plasminogen activator
Updated on 24-November-2019
References
- ↑ Sodeinde OA, Sample AK, Brubaker RR, Goguen JD. Plasminogen activator/coagulase gene of Yersinia pestis is responsible for degradation of plasmid-encoded outer membrane proteins. Infect Immun. 1988 Oct;56(10):2749-52. PMID:2843471
- ↑ . Tissue plasminogen activator for acute ischemic stroke. The National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group. N Engl J Med. 1995 Dec 14;333(24):1581-7. PMID:7477192 doi:http://dx.doi.org/10.1056/NEJM199512143332401