This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
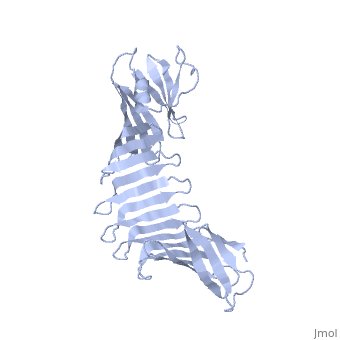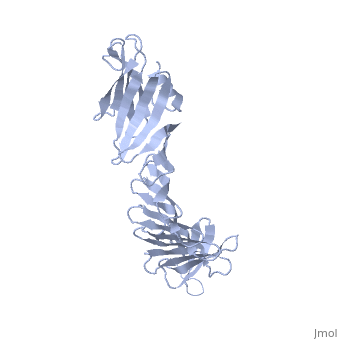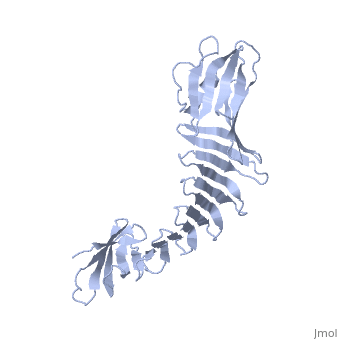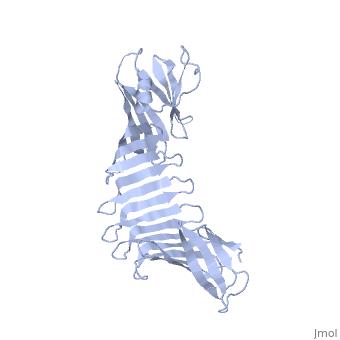
2af5
From Proteopedia
(Difference between revisions)
| (13 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
| - | [[Image:2af5.gif|left|200px]] | ||
| - | + | ==2.5A X-ray Structure of Engineered OspA protein== | |
| - | + | <StructureSection load='2af5' size='340' side='right'caption='[[2af5]], [[Resolution|resolution]] 2.50Å' scene=''> | |
| - | + | == Structural highlights == | |
| - | + | <table><tr><td colspan='2'>[[2af5]] is a 1 chain structure with sequence from [https://en.wikipedia.org/wiki/Borreliella_burgdorferi Borreliella burgdorferi]. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=2AF5 OCA]. For a <b>guided tour on the structure components</b> use [https://proteopedia.org/fgij/fg.htm?mol=2AF5 FirstGlance]. <br> | |
| - | + | </td></tr><tr id='method'><td class="sblockLbl"><b>[[Empirical_models|Method:]]</b></td><td class="sblockDat" id="methodDat">X-ray diffraction, [[Resolution|Resolution]] 2.5Å</td></tr> | |
| - | + | <tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[https://proteopedia.org/fgij/fg.htm?mol=2af5 FirstGlance], [http://oca.weizmann.ac.il/oca-bin/ocaids?id=2af5 OCA], [https://pdbe.org/2af5 PDBe], [https://www.rcsb.org/pdb/explore.do?structureId=2af5 RCSB], [https://www.ebi.ac.uk/pdbsum/2af5 PDBsum], [https://prosat.h-its.org/prosat/prosatexe?pdbcode=2af5 ProSAT]</span></td></tr> | |
| - | + | </table> | |
| - | + | == Function == | |
| - | + | [https://www.uniprot.org/uniprot/OSPA_BORBU OSPA_BORBU] | |
| - | + | == Evolutionary Conservation == | |
| - | + | [[Image:Consurf_key_small.gif|200px|right]] | |
| - | + | Check<jmol> | |
| - | + | <jmolCheckbox> | |
| - | + | <scriptWhenChecked>; select protein; define ~consurf_to_do selected; consurf_initial_scene = true; script "/wiki/ConSurf/af/2af5_consurf.spt"</scriptWhenChecked> | |
| - | == | + | <scriptWhenUnchecked>script /wiki/extensions/Proteopedia/spt/initialview01.spt</scriptWhenUnchecked> |
| + | <text>to colour the structure by Evolutionary Conservation</text> | ||
| + | </jmolCheckbox> | ||
| + | </jmol>, as determined by [http://consurfdb.tau.ac.il/ ConSurfDB]. You may read the [[Conservation%2C_Evolutionary|explanation]] of the method and the full data available from [http://bental.tau.ac.il/new_ConSurfDB/main_output.php?pdb_ID=2af5 ConSurf]. | ||
| + | <div style="clear:both"></div> | ||
| + | <div style="background-color:#fffaf0;"> | ||
| + | == Publication Abstract from PubMed == | ||
Although the beta-rich self-assemblies are a major structural class for polypeptides and the focus of intense research, little is known about their atomic structures and dynamics due to their insoluble and noncrystalline nature. We developed a protein engineering strategy that captures a self-assembly segment in a water-soluble molecule. A predefined number of self-assembling peptide units are linked, and the beta-sheet ends are capped to prevent aggregation, which yields a mono-dispersed soluble protein. We tested this strategy by using Borrelia outer surface protein (OspA) whose single-layer beta-sheet located between two globular domains consists of two beta-hairpin units and thus can be considered as a prototype of self-assembly. We constructed self-assembly mimics of different sizes and determined their atomic structures using x-ray crystallography and NMR spectroscopy. Highly regular beta-sheet geometries were maintained in these structures, and peptide units had a nearly identical conformation, supporting the concept that a peptide in the regular beta-geometry is primed for self-assembly. However, we found small but significant differences in the relative orientation between adjacent peptide units in terms of beta-sheet twist and bend, suggesting their inherent flexibility. Modeling shows how this conformational diversity, when propagated over a large number of peptide units, can lead to a substantial degree of nanoscale polymorphism of self-assemblies. | Although the beta-rich self-assemblies are a major structural class for polypeptides and the focus of intense research, little is known about their atomic structures and dynamics due to their insoluble and noncrystalline nature. We developed a protein engineering strategy that captures a self-assembly segment in a water-soluble molecule. A predefined number of self-assembling peptide units are linked, and the beta-sheet ends are capped to prevent aggregation, which yields a mono-dispersed soluble protein. We tested this strategy by using Borrelia outer surface protein (OspA) whose single-layer beta-sheet located between two globular domains consists of two beta-hairpin units and thus can be considered as a prototype of self-assembly. We constructed self-assembly mimics of different sizes and determined their atomic structures using x-ray crystallography and NMR spectroscopy. Highly regular beta-sheet geometries were maintained in these structures, and peptide units had a nearly identical conformation, supporting the concept that a peptide in the regular beta-geometry is primed for self-assembly. However, we found small but significant differences in the relative orientation between adjacent peptide units in terms of beta-sheet twist and bend, suggesting their inherent flexibility. Modeling shows how this conformational diversity, when propagated over a large number of peptide units, can lead to a substantial degree of nanoscale polymorphism of self-assemblies. | ||
| - | + | Atomic structures of peptide self-assembly mimics.,Makabe K, McElheny D, Tereshko V, Hilyard A, Gawlak G, Yan S, Koide A, Koide S Proc Natl Acad Sci U S A. 2006 Nov 21;103(47):17753-8. Epub 2006 Nov 8. PMID:17093048<ref>PMID:17093048</ref> | |
| - | + | ||
| - | + | From MEDLINE®/PubMed®, a database of the U.S. National Library of Medicine.<br> | |
| - | + | </div> | |
| - | + | <div class="pdbe-citations 2af5" style="background-color:#fffaf0;"></div> | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ==See Also== | |
| + | *[[Outer surface protein|Outer surface protein]] | ||
| + | == References == | ||
| + | <references/> | ||
| + | __TOC__ | ||
| + | </StructureSection> | ||
| + | [[Category: Borreliella burgdorferi]] | ||
| + | [[Category: Large Structures]] | ||
| + | [[Category: Hilyard A]] | ||
| + | [[Category: Koide A]] | ||
| + | [[Category: Koide S]] | ||
| + | [[Category: Makabe K]] | ||
| + | [[Category: Mcelheny D]] | ||
| + | [[Category: Tereshko V]] | ||
Current revision
2.5A X-ray Structure of Engineered OspA protein
| |||||||||||