This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Heat Shock Proteins
From Proteopedia
(Difference between revisions)
| (6 intermediate revisions not shown.) | |||
| Line 2: | Line 2: | ||
== Function == | == Function == | ||
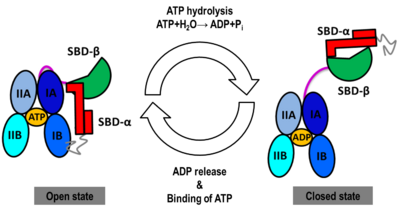
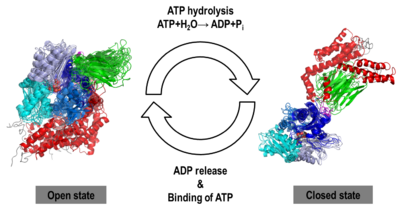
| - | [[Heat Shock Proteins]] (Hsp). The Hsp are proteins which are expressed when cells are exposed to stresses like heat. Hsp60, Hsp70, Hsp90 etc. are named according to their molecular weights. Their function is to provide protection from stress<ref>PMID:18432918</ref>. Hsp contains a nucleotide-binding domain (NBD), a substrate-binding domain (SBD) and C-terminal lid domain (lid). In ''E. coli'' there are additional Hsp – HslV (19kD), HslJ and HslU (50kD). The heat-inducible Hsp is called '''heat shock cognate protein''' (Hsc). '''Hsp70''' from ''E. coli'' is called '''DnaK'''. '''Hsp90B1''' is also called '''endoplasmin''' and '''grp94'''. | + | [[Heat Shock Proteins]] (Hsp). The Hsp are proteins which are expressed when cells are exposed to stresses like heat. Hsp60, Hsp70, Hsp90 etc. are named according to their molecular weights. Their function is to provide protection from stress<ref>PMID:18432918</ref>. Hsp contains a nucleotide-binding domain (NBD), a substrate-binding domain (SBD) and C-terminal lid domain (lid). In ''E. coli'' there are additional Hsp – HslV (19kD), HslJ and HslU (50kD). The heat-inducible Hsp is called '''heat shock cognate protein''' (Hsc). '''Hsp70''' from ''E. coli'' is called '''DnaK'''. '''Hsp70 family protein 5''' or '''endoplasmic reticulum chaperone BIP''' is located in the lumen of the ER and its synthesis is markedly induced under conditions that lead to the accumulation of unfolded polypeptides in the ER<ref>apmid;10597629</ref> '''Hsp90B1''' is also called '''endoplasmin''' and '''grp94'''. '''DegP''' is a dual-function chaperon and protease. It degrades peripasmic proteins under stress conditions<ref>PMID:31738379</ref>. |
See also<br /> | See also<br /> | ||
* [[Chaperones]]<br /> | * [[Chaperones]]<br /> | ||
* [[Hsp100]]<br /> | * [[Hsp100]]<br /> | ||
* [[Hsp40]] | * [[Hsp40]] | ||
| + | * [[Hsp70]] | ||
* [[Molecular Playground/DnaK]] for Hsp70 in ''E. coli'' | * [[Molecular Playground/DnaK]] for Hsp70 in ''E. coli'' | ||
* [[Molecular Playground/sHSP]] for small Hsp | * [[Molecular Playground/sHSP]] for small Hsp | ||
| + | * [[Molecular Playground/Hsp70-Hsp90]] | ||
== Conformational dynamics of full-length inducible human Hsp70 derived from microsecond molecular dynamics simulations in explicit solvent <ref>doi 10.1080/07391102.2012.726190</ref> == | == Conformational dynamics of full-length inducible human Hsp70 derived from microsecond molecular dynamics simulations in explicit solvent <ref>doi 10.1080/07391102.2012.726190</ref> == | ||
Current revision
| |||||||||||
References
- ↑ Li Z, Srivastava P. Heat-shock proteins. Curr Protoc Immunol. 2004 Feb;Appendix 1:Appendix 1T. doi:, 10.1002/0471142735.ima01ts58. PMID:18432918 doi:http://dx.doi.org/10.1002/0471142735.ima01ts58
- ↑ apmid;10597629
- ↑ Zhang S, Cheng Y, Ma J, Wang Y, Chang Z, Fu X. Degp degrades a wide range of substrate proteins in Escherichia coli under stress conditions. Biochem J. 2019 Dec 12;476(23):3549-3564. PMID:31738379 doi:10.1042/BCJ20190446
- ↑ Nicolai A, Delarue P, Senet P. Conformational dynamics of full-length inducible human Hsp70 derived from microsecond molecular dynamics simulations in explicit solvent. J Biomol Struct Dyn. 2012 Oct 17. PMID:23075261 doi:10.1080/07391102.2012.726190