This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
5llk
From Proteopedia
(Difference between revisions)
| Line 3: | Line 3: | ||
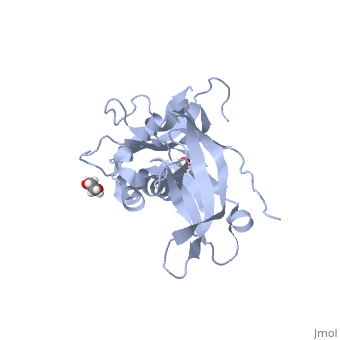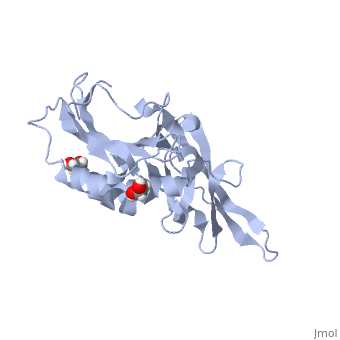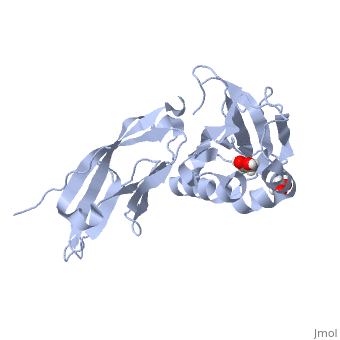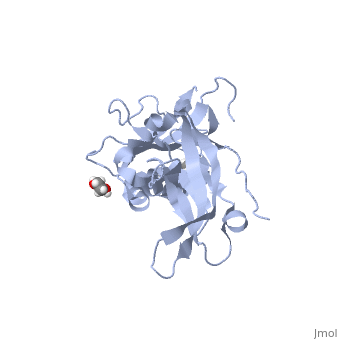
<StructureSection load='5llk' size='340' side='right'caption='[[5llk]], [[Resolution|resolution]] 1.80Å' scene=''> | <StructureSection load='5llk' size='340' side='right'caption='[[5llk]], [[Resolution|resolution]] 1.80Å' scene=''> | ||
== Structural highlights == | == Structural highlights == | ||
| - | <table><tr><td colspan='2'>[[5llk]] is a 1 chain structure. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=5LLK OCA]. For a <b>guided tour on the structure components</b> use [ | + | <table><tr><td colspan='2'>[[5llk]] is a 1 chain structure with sequence from [https://en.wikipedia.org/wiki/Homo_sapiens Homo sapiens]. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=5LLK OCA]. For a <b>guided tour on the structure components</b> use [https://proteopedia.org/fgij/fg.htm?mol=5LLK FirstGlance]. <br> |
| - | </td></tr><tr id='ligand'><td class="sblockLbl"><b>[[Ligand|Ligands:]]</b></td><td class="sblockDat" id="ligandDat"><scene name='pdbligand=EDO:1,2-ETHANEDIOL'>EDO</scene></td></tr> | + | </td></tr><tr id='method'><td class="sblockLbl"><b>[[Empirical_models|Method:]]</b></td><td class="sblockDat" id="methodDat">X-ray diffraction, [[Resolution|Resolution]] 1.8Å</td></tr> |
| - | <tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[ | + | <tr id='ligand'><td class="sblockLbl"><b>[[Ligand|Ligands:]]</b></td><td class="sblockDat" id="ligandDat"><scene name='pdbligand=EDO:1,2-ETHANEDIOL'>EDO</scene></td></tr> |
| + | <tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[https://proteopedia.org/fgij/fg.htm?mol=5llk FirstGlance], [http://oca.weizmann.ac.il/oca-bin/ocaids?id=5llk OCA], [https://pdbe.org/5llk PDBe], [https://www.rcsb.org/pdb/explore.do?structureId=5llk RCSB], [https://www.ebi.ac.uk/pdbsum/5llk PDBsum], [https://prosat.h-its.org/prosat/prosatexe?pdbcode=5llk ProSAT]</span></td></tr> | ||
</table> | </table> | ||
== Disease == | == Disease == | ||
| - | [ | + | [https://www.uniprot.org/uniprot/DAG1_HUMAN DAG1_HUMAN] Defects in DAG1 are the cause of muscular dystrophy-dystroglycanopathy limb-girdle type C7 (MDDGC7) [MIM:[https://omim.org/entry/613818 613818]. An autosomal recessive muscular dystrophy showing onset in early childhood, and associated with mental retardation without structural brain anomalies. Note=MDDGC7 is caused by DAG1 mutations that interfere with normal post-translational processing, resulting in defective DAG1 glycosylation and impaired interactions with extracellular-matrix components. Other muscular dystrophy-dystroglycanopathies are caused by defects in enzymes involved in protein O-glycosylation.<ref>PMID:21388311</ref> |
== Function == | == Function == | ||
| - | [ | + | [https://www.uniprot.org/uniprot/DAG1_HUMAN DAG1_HUMAN] The dystroglycan complex is involved in a number of processes including laminin and basement membrane assembly, sarcolemmal stability, cell survival, peripheral nerve myelination, nodal structure, cell migration, and epithelial polarization.<ref>PMID:9851927</ref> <ref>PMID:11724572</ref> <ref>PMID:16254364</ref> <ref>PMID:17360738</ref> Alpha-dystroglycan is an extracellular peripheral glycoprotein that acts as a receptor for both extracellular matrix proteins containing laminin-G domains, and for certain adenoviruses. Receptor for laminin-2 (LAMA2) and agrin in peripheral nerve Schwann cells. Also acts as a receptor for M.leprae in peripheral nerve Schwann cells but only in the presence of the G-domain of LAMA2, and for lymphocytic choriomeningitis virus, Old World Lassa fever virus, and clade C New World arenaviruses.<ref>PMID:9851927</ref> <ref>PMID:11724572</ref> <ref>PMID:16254364</ref> <ref>PMID:17360738</ref> Beta-dystroglycan is a transmembrane protein that plays important roles in connecting the extracellular matrix to the cytoskeleton. Acts as a cell adhesion receptor in both muscle and non-muscle tissues. Receptor for both DMD and UTRN and, through these interactions, scaffolds axin to the cytoskeleton. Also functions in cell adhesion-mediated signaling and implicated in cell polarity.<ref>PMID:9851927</ref> <ref>PMID:11724572</ref> <ref>PMID:16254364</ref> <ref>PMID:17360738</ref> |
<div style="background-color:#fffaf0;"> | <div style="background-color:#fffaf0;"> | ||
== Publication Abstract from PubMed == | == Publication Abstract from PubMed == | ||
| Line 27: | Line 28: | ||
__TOC__ | __TOC__ | ||
</StructureSection> | </StructureSection> | ||
| + | [[Category: Homo sapiens]] | ||
[[Category: Large Structures]] | [[Category: Large Structures]] | ||
| - | [[Category: Bigotti | + | [[Category: Bigotti MG]] |
| - | [[Category: Bozzi | + | [[Category: Bozzi M]] |
| - | [[Category: Brancaccio | + | [[Category: Brancaccio A]] |
| - | [[Category: Cassetta | + | [[Category: Cassetta A]] |
| - | [[Category: Covaceuszach | + | [[Category: Covaceuszach S]] |
| - | [[Category: Konarev | + | [[Category: Konarev PV]] |
| - | [[Category: Lamba | + | [[Category: Lamba D]] |
| - | [[Category: Sciandra | + | [[Category: Sciandra F]] |
| - | + | ||
| - | + | ||
| - | + | ||
Current revision
Crystal structure of human alpha-dystroglycan
| |||||||||||