This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Methane monooxygenase
From Proteopedia
(Difference between revisions)
| (5 intermediate revisions not shown.) | |||
| Line 3: | Line 3: | ||
== Function == | == Function == | ||
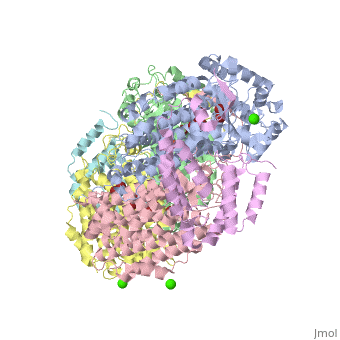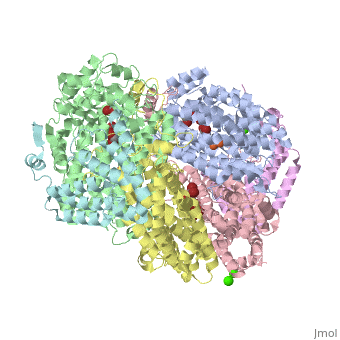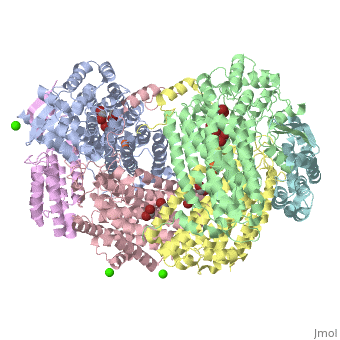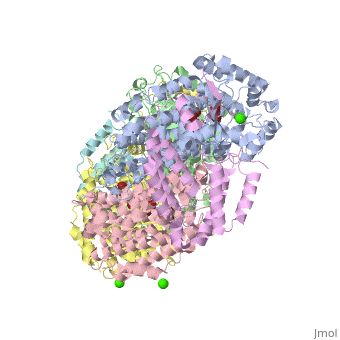
| - | '''Methane monooxygenase''' (MMO) catalyzes the oxidation of the C-H bond of methane and other alkanes. MMO catalyzes the conversion of CH<sub>4</sub> and O<sub>2</sub> to CH<sub>3</sub>OH and H<sub>2</sub>O using NADPH as reducing agent. The '''soluble MMO (sMMO)''' has Fe-O-Fe in its active site<ref>PMID:7826011</ref> while the '''particulate MMO (pMMO)''' has Cu in it<ref>PMID:21419924</ref>. MMO is composed of 3 subunits.<br /> | + | '''Methane monooxygenase''' or '''Methane monooxygenase hydroxylase''' (MMO) catalyzes the oxidation of the C-H bond of methane and other alkanes. MMO catalyzes the conversion of CH<sub>4</sub> and O<sub>2</sub> to CH<sub>3</sub>OH and H<sub>2</sub>O using NADPH as reducing agent. The '''soluble MMO (sMMO)''' has Fe-O-Fe in its active site<ref>PMID:7826011</ref> while the '''particulate MMO (pMMO)''' has Cu in it<ref>PMID:21419924</ref>. MMO is composed of 3 subunits.<br /> |
* Subunit α is the hydroxylase (MMOH) and contains the di-iron active site.<br /> | * Subunit α is the hydroxylase (MMOH) and contains the di-iron active site.<br /> | ||
* Subunit β is a reductase which contains the co-factors FAD and 2Fe-2S complex.<br /> | * Subunit β is a reductase which contains the co-factors FAD and 2Fe-2S complex.<br /> | ||
| Line 9: | Line 9: | ||
MMO is found in methanotropic bacteria. | MMO is found in methanotropic bacteria. | ||
| - | + | == Methane monooxygenase 3D structures== | |
| - | + | [[Methane monooxygenase 3D structures]] | |
| - | + | </StructureSection> | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
== References == | == References == | ||
Current revision
| |||||||||||
References
- ↑ Lipscomb JD. Biochemistry of the soluble methane monooxygenase. Annu Rev Microbiol. 1994;48:371-99. PMID:7826011 doi:http://dx.doi.org/10.1146/annurev.mi.48.100194.002103
- ↑ Miyaji A. Particulate methane monooxygenase from Methylosinus trichosporium OB3b. Methods Enzymol. 2011;495:211-25. doi: 10.1016/B978-0-12-386905-0.00014-0. PMID:21419924 doi:http://dx.doi.org/10.1016/B978-0-12-386905-0.00014-0