This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Penicillin-binding protein
From Proteopedia
(Difference between revisions)
| (2 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
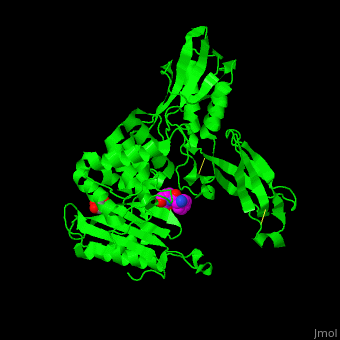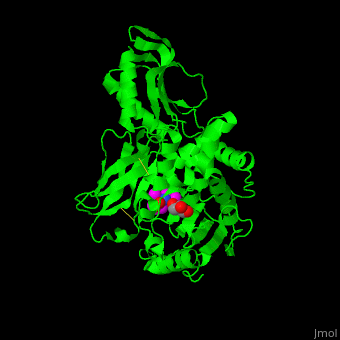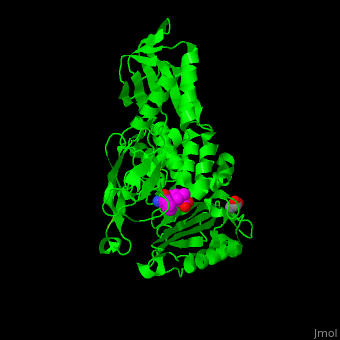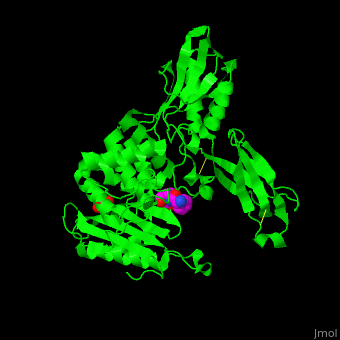
<StructureSection load='' size='350' side='right' scene='47/478544/Cv/1' caption='E. coli PBP 4 complex with the antibiotic ampicillin and glycerol [[2ex6]]'> | <StructureSection load='' size='350' side='right' scene='47/478544/Cv/1' caption='E. coli PBP 4 complex with the antibiotic ampicillin and glycerol [[2ex6]]'> | ||
== Function == | == Function == | ||
| + | Penicillin-binding proteins (PBPs) are a group of proteins that are characterized by their affinity for and binding of penicillin. They are a normal constituent of many bacteria; the name just reflects the way by which the protein was discovered. All β-lactam antibiotics (except for tabtoxinine-β-lactam, which inhibits glutamine synthetase) bind to PBPs, which are essential for bacterial cell wall synthesis. PBPs are members of a subgroup of enzymes called transpeptidases. Specifically, PBPs are DD-transpeptidases. See also [https://en.wikipedia.org/wiki/Penicillin-binding_proteins Penicillin-binding proteins]. | ||
| - | '''Penicillin-binding protein''' or '''peptidoglycan d,d-transpeptidase''' (PBP) is a bacterial protein which binds antibiotics. There are several PBPs in each organism. PBPs are involved in the synthesis of bacterial cell wall<ref>PMID:1103132</ref>. The PBP are classified to high-molecular weight and low-molecular weight groups. '''PBP 3''' is also named '''FtsI'''. | + | '''Penicillin-binding protein''' or '''peptidoglycan d,d-transpeptidase''' or '''D-alanyl-D-alanine carboxypeptidase''' (PBP) is a bacterial protein which binds antibiotics. There are several PBPs in each organism. PBPs are involved in the synthesis of bacterial cell wall<ref>PMID:1103132</ref>. The PBP are classified to high-molecular weight and low-molecular weight groups. '''PBP 3''' is also named '''FtsI'''. |
| + | See also: | ||
| + | *[[DD-transpeptidase (Hebrew)]]. | ||
| + | *[[Sandbox 126|Penicillin-binding proteins and antibiotics]] | ||
For ''Mycobacterium tuberculosis'' PBP complex with penicillin see [[Mycobacterium Tuberculosis Transpeptidase Domain]]. | For ''Mycobacterium tuberculosis'' PBP complex with penicillin see [[Mycobacterium Tuberculosis Transpeptidase Domain]]. | ||
Current revision
| |||||||||||
References
- ↑ Spratt BG. Distinct penicillin binding proteins involved in the division, elongation, and shape of Escherichia coli K12. Proc Natl Acad Sci U S A. 1975 Aug;72(8):2999-3003. PMID:1103132
- ↑ Beadle BM, Nicholas RA, Shoichet BK. Interaction energies between beta-lactam antibiotics and E. coli penicillin-binding protein 5 by reversible thermal denaturation. Protein Sci. 2001 Jun;10(6):1254-9. PMID:11369864 doi:http://dx.doi.org/10.1110/ps.52001
- ↑ Kishida H, Unzai S, Roper DI, Lloyd A, Park SY, Tame JR. Crystal structure of penicillin binding protein 4 (dacB) from Escherichia coli, both in the native form and covalently linked to various antibiotics. Biochemistry. 2006 Jan 24;45(3):783-92. PMID:16411754 doi:10.1021/bi051533t