This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
DNA Polymerase Theta
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
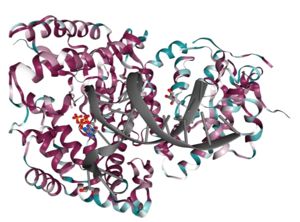
| - | <StructureSection load='4X0P' size='350' side='right' caption='DNA polymerase theta polymerase domain in complex with double-stranded DNA substrate and incoming ddATP ( | + | <StructureSection load='4X0P' size='350' side='right' caption='DNA polymerase theta polymerase domain in complex with double-stranded DNA substrate and incoming ddATP (4x0p)' scene=''> |
==Structural Description== | ==Structural Description== | ||
| - | The polymerase domain of DNA polymerase theta (<scene name='84/842971/Qm1/1'>QM1</scene>) is a member of the A-family of polymerases which includes Pol I <ref>PMID: 8469987</ref> and T7 DNA polymerase <ref>PMID: 9440688</ref>. A-family polymerases contain <scene name='84/842971/A-family_motifs/1'>6 conserved motifs</scene>. These motifs allow for identification of A-family polymerases based on their amino acid sequence. In addition to the A-family motifs, the <scene name='84/842971/Finger-thumb-palm/1'>finger-palm-thumb</scene> subdomains that are typical of many polymerases are also conserved in QM1. The <scene name='84/842971/Fingers/1'>fingers subdomain</scene> coordinate the <scene name='84/842971/Dna/1'>DNA</scene> template and the <scene name='84/842971/Dds/1'>incoming nucleotide</scene> while the <scene name='84/842971/Palm/1'>palm subdomain</scene> performs catalysis and the <scene name='84/842971/Thumb/1'>thumb subdomain</scene> contacts the double stranded DNA. Within these subdomains exist disordered structural inserts that are not present in bacterial homologs. These inserts impart unique properties to QM1;<scene name='84/842971/Insert1/1'> insert 1</scene> in the thumb increases processivity while inserts <scene name='84/842971/Insert2/1'>2</scene> and <scene name='84/842971/Insert3/1'>3</scene> facilitate the ability of QM1 to bypass <scene name='84/842971/Thf/1'>DNA lesions</scene> that typically block synthesis by other A-family polymerases. This error prone synthesis makes QM1 more functionally similar to Y-family translesion polymerases. | + | The polymerase domain of '''DNA polymerase theta''' (<scene name='84/842971/Qm1/1'>QM1</scene>) is a member of the A-family of polymerases which includes Pol I <ref>PMID: 8469987</ref> and T7 DNA polymerase <ref>PMID: 9440688</ref>. A-family polymerases contain <scene name='84/842971/A-family_motifs/1'>6 conserved motifs</scene>. These motifs allow for identification of A-family polymerases based on their amino acid sequence. In addition to the A-family motifs, the <scene name='84/842971/Finger-thumb-palm/1'>finger-palm-thumb</scene> subdomains that are typical of many polymerases are also conserved in QM1. The <scene name='84/842971/Fingers/1'>fingers subdomain</scene> coordinate the <scene name='84/842971/Dna/1'>DNA</scene> template and the <scene name='84/842971/Dds/1'>incoming nucleotide</scene> while the <scene name='84/842971/Palm/1'>palm subdomain</scene> performs catalysis and the <scene name='84/842971/Thumb/1'>thumb subdomain</scene> contacts the double stranded DNA. Within these subdomains exist disordered structural inserts that are not present in bacterial homologs. These inserts impart unique properties to QM1;<scene name='84/842971/Insert1/1'> insert 1</scene> in the thumb increases processivity while inserts <scene name='84/842971/Insert2/1'>2</scene> and <scene name='84/842971/Insert3/1'>3</scene> facilitate the ability of QM1 to bypass <scene name='84/842971/Thf/1'>DNA lesions</scene> that typically block synthesis by other A-family polymerases. This error prone synthesis makes QM1 more functionally similar to Y-family translesion polymerases. |
Beyond the canonical polymerase folds, QM1 also possesses a vestigial <scene name='84/842971/Exonuclease/1'>exonuclease</scene> domain that no longer performs proof-reading activities. This domain also contains <scene name='84/842971/Exo_1_2/1'>two structural inserts</scene> that are thought to contact the more distant helicase-like and central domains of full-length polymerase theta (see Function). | Beyond the canonical polymerase folds, QM1 also possesses a vestigial <scene name='84/842971/Exonuclease/1'>exonuclease</scene> domain that no longer performs proof-reading activities. This domain also contains <scene name='84/842971/Exo_1_2/1'>two structural inserts</scene> that are thought to contact the more distant helicase-like and central domains of full-length polymerase theta (see Function). | ||
Current revision
| |||||||||||