This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Madison Unger/Sandbox 1
From Proteopedia
< User:Madison Unger(Difference between revisions)
| (12 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
=Human Acyl-Coenzyme A= | =Human Acyl-Coenzyme A= | ||
| - | <StructureSection load='6l47' size='340' frame='true' side='right' caption='Functioning dimer of ACAT' scene='87/877507/Dimer/ | + | <StructureSection load='6l47' size='340' frame='true' side='right' caption='Functioning dimer of ACAT' scene='87/877507/Dimer/7'> |
== Introduction == | == Introduction == | ||
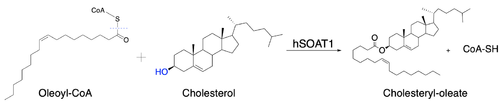
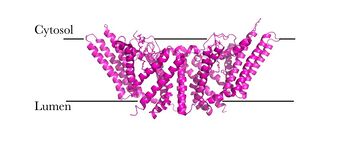
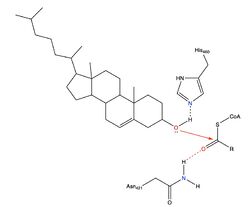
| - | [[Image:Newmechbigwords.png|500px|right|thumb|Figure 1: Overall reaction scheme of ACAT]] Acyl-coenzyme A: cholesterol acyltransferase <scene name='87/877507/Dimer/ | + | [[Image:Newmechbigwords.png|500px|right|thumb|Figure 1: Overall reaction scheme of ACAT]] Acyl-coenzyme A: cholesterol acyltransferase <scene name='87/877507/Dimer/7'>(ACAT)</scene>, also known as Human Sterol O-acyltransferase (hSOAT) is an enzyme that catalyzes the reaction between long chain [http://en.wikipedia.org/wiki/Long-chain-fatty-acid%E2%80%94CoA_ligase fatty acyl CoA], the <scene name='87/877507/Acyl-coa_surface/4'>naturally-occurring substrate</scene>, and intracellular [http://en.wikipedia.org/wiki/Cholesterol cholesterol] to form the more hydrophobic cholesteryl ester for cholesterol storage (Fig.1). [http://en.wikipedia.org/wiki/Cholesteryl_ester Cholesteryl ester] is the primary form of how cholesterol is stored in multiple types of cells and transported through the circulatory system. ACAT is an endoplasmic reticulum membrane protein with a specific orientation (Fig. 2). ACAT is a part of the [http://en.wikipedia.org/wiki/MBOAT MBOAT] (membrane-bound O-acyltransferase) family, which also includes acyl-coenzyme A: <scene name='87/877507/Dgat/4'>diacylglycerol acyltransferase</scene> ([http://en.wikipedia.org/wiki/Diglyceride_acyltransferase DGAT]) <ref>PMID:32433610</ref> and <scene name='87/877507/Goat/1'>ghrelin O-acyltransferase</scene>([http://en.wikipedia.org/wiki/Ghrelin_O-acyltransferase GOAT]). [[Image:Cytosol lumen pic.jpg|360px|right|thumb|Figure 2: Orientation showing the cytosolic and lumen sides of the dimer]]<ref>PMID:19356147</ref> |
There have been two ACAT [http://en.wikipedia.org/wiki/Protein_isoform isoforms] discovered in mammals, ACAT1<ref name="Qian">PMID:32433614</ref> and ACAT2<ref name="Cases">PMID:9756919</ref>, and they are predominantly located in different parts of the body. ACAT1 is mainly found in the liver, kidneys, adrenal glands and macrophages, whereas ACAT2 is found only in the intestines and liver. | There have been two ACAT [http://en.wikipedia.org/wiki/Protein_isoform isoforms] discovered in mammals, ACAT1<ref name="Qian">PMID:32433614</ref> and ACAT2<ref name="Cases">PMID:9756919</ref>, and they are predominantly located in different parts of the body. ACAT1 is mainly found in the liver, kidneys, adrenal glands and macrophages, whereas ACAT2 is found only in the intestines and liver. | ||
| Line 8: | Line 8: | ||
== Structure == | == Structure == | ||
=== Overall Structure === | === Overall Structure === | ||
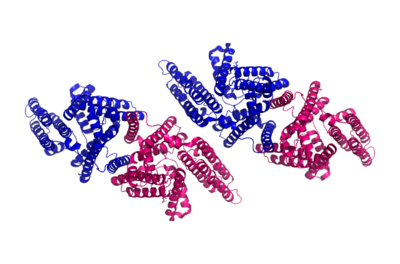
| - | ACAT is a tetramer composed of a [http://en.wikipedia.org/wiki/Protein_dimer dimer] of a dimer, but is able to perform its function solely as a dimer (Fig. 3). [[Image:Tetramer_dimer_of_dimer.png|400px|left|thumb|Figure 3: Tetrameric dimer of dimer for ACAT]]There are <scene name='87/877508/9_transmembrane_helices/ | + | ACAT is a tetramer composed of a [http://en.wikipedia.org/wiki/Protein_dimer dimer] of a dimer, but is able to perform its function solely as a dimer (Fig. 3). [[Image:Tetramer_dimer_of_dimer.png|400px|left|thumb|Figure 3: Tetrameric dimer of dimer for ACAT]]There are <scene name='87/877508/9_transmembrane_helices/5'>nine transmembrane helices</scene> in each domain which create a tunnel for the active site. There are also three helices found on the intracellular side (IH1, IH2, and IH3) and one helix on the extracellular side (EH1). The active site contains three tunnels – the transmembrane tunnel for cholesterol entrance, the cytosolic tunnel for acyl-CoA entrance, and the lumen tunnel for cholesterol ester exit. ACAT also has an amino-terminal cytosolic domain (NTD) that is important for tetramerization of this protein.<ref name= "Cases" /> |
=== Dimer-Dimer Interactions === | === Dimer-Dimer Interactions === | ||
| - | The two dimers make limited contact within the membrane through an interface that has <scene name='87/877508/Dimer_interface/ | + | The two dimers make limited contact within the membrane through an interface that has <scene name='87/877508/Dimer_interface/2'>hydrophobic residues</scene> in between the two protomers <ref name="Qian" />. Between the two protomers in each dimer, Van der Waals interactions occur between TM1 of one [http://en.wikipedia.org/wiki/Protomer protomer] and the lumenal TM6 and the cytosolic TM9 of the other protomer. |
===Tunnels=== | ===Tunnels=== | ||
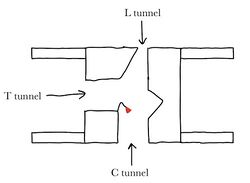
| - | The nine transmembrane segments create a <scene name='87/877507/C_tunnel_final1/ | + | The nine transmembrane segments create a <scene name='87/877507/C_tunnel_final1/4'>cytosolic tunnel</scene> and a <scene name='87/877507/T-tunnel_april_19/6'>transmembrane tunnel</scene> that meet at the site of catalysis (active site). Acyl-coenzyme A enters the active site though the cytosolic tunnel, and cholesterol enters through the transmembrane tunnel (Fig.4). These then meet in the catalytic site to react and form the cholesteryl ester.[[Image:drawntunnels.jpg|250px|right|thumb|Figure 4: Transmembrane, cytosolic, and lumen tunnels drawn with catalytic His460 (shown as red object) at the convergence of three tunnels]] When exiting the catalytic site, the free CoA is able to release through the cytosolic tunnel to the cytosol, and the cholesterol ester is able to release through the transmembrane tunnel to the membrane or through the lumen tunnel to the lumen. Certain residues that line the transmembrane tunnel are important in ACAT activity (E259, E263, R262, P304, L306, V423, V424, M265, I261, and H460)<ref name="Qian" />. |
=== Active Site/Important Residues === | === Active Site/Important Residues === | ||
| - | An important residue in the ACAT active site is <scene name='87/877507/H460_final_2/ | + | An important residue in the ACAT active site is <scene name='87/877507/H460_final_2/4'>His460</scene>, a Histidine, which is located where the tunnels converge. It is thought that His460 is located on TM7<ref name = "Qian" />. When converting to a cholesteryl ester, the His460 acts as a catalytic base that deprotonates the cholesterol. An asparagine <scene name='87/877507/Asn241_finalhydrogenbond/3'>Asn421</scene> is another important residue in the reaction that is able to form a hydrogen bond with acyl-CoA for stabilization. Additionally, a <scene name='87/877507/Acyl-coa_surface/4'>naturally-occurring substrate</scene> can be found in the active site of ACAT and replaced by cholesterol for synthesis of cholesteryl ester. |
== Proposed Mechanism == | == Proposed Mechanism == | ||
Due to limited high-resolution structural representations of ACAT, its mechanism remains ambiguous. [[Image:Thenewacatmech2.jpg|250px|left|thumb|Figure 5: Mechanism for ACAT proposed by Qian et al.<ref name = "Qian" />]] However, the general mechanism involving the substrates and products of ACAT is understood<ref name="Guan">PMID:32424158</ref>. In this reaction, [http://en.wikipedia.org/wiki/Stearoyl-CoA_9-desaturase oleoyl-CoA] and cholesterol are the reactants and they undergo the reaction catalyzed by ACAT to form cholesteryl-oleate which is used as a storage form of cholesterol. The hydroxyl group on cholesterol is deprotonated, then attacks the [http://en.wikipedia.org/wiki/Thioester thioester] bond of oleoyl-CoA, kicking off CoA-SH as a leaving group. | Due to limited high-resolution structural representations of ACAT, its mechanism remains ambiguous. [[Image:Thenewacatmech2.jpg|250px|left|thumb|Figure 5: Mechanism for ACAT proposed by Qian et al.<ref name = "Qian" />]] However, the general mechanism involving the substrates and products of ACAT is understood<ref name="Guan">PMID:32424158</ref>. In this reaction, [http://en.wikipedia.org/wiki/Stearoyl-CoA_9-desaturase oleoyl-CoA] and cholesterol are the reactants and they undergo the reaction catalyzed by ACAT to form cholesteryl-oleate which is used as a storage form of cholesterol. The hydroxyl group on cholesterol is deprotonated, then attacks the [http://en.wikipedia.org/wiki/Thioester thioester] bond of oleoyl-CoA, kicking off CoA-SH as a leaving group. | ||
| - | However, Qian et al.<ref name="Qian" /> proposed a mechanism involving the important residues <scene name='87/877507/H460_final_2/ | + | However, Qian et al.<ref name="Qian" /> proposed a mechanism involving the important residues <scene name='87/877507/H460_final_2/4'>His460</scene> and <scene name='87/877507/Asn241_finalhydrogenbond/3'>Asn241</scene>. In this mechanism, His460 acts as a general base to deprotonate the hydroxyl group on cholesterol, activating it as a [http://en.wikipedia.org/wiki/Nucleophile nucleophile]. Then, Asn421 possibly forms a hydrogen bond with oleoyl-CoA to stabilize the reaction (Fig. 5). |
Current revision
Human Acyl-Coenzyme A
| |||||||||||
References
- ↑ Wang L, Qian H, Nian Y, Han Y, Ren Z, Zhang H, Hu L, Prasad BVV, Laganowsky A, Yan N, Zhou M. Structure and mechanism of human diacylglycerol O-acyltransferase 1. Nature. 2020 May;581(7808):329-332. doi: 10.1038/s41586-020-2280-2. Epub 2020 May, 13. PMID:32433610 doi:http://dx.doi.org/10.1038/s41586-020-2280-2
- ↑ Moorthy PS, Neelagandan K, Balasubramanian M, Ponnuswamy MN. Purification, Crystallization and Preliminary X-Ray Diffraction Studies on Goat (Capra hircus) Hemoglobin - A Low Oxygen Affinity Species. Protein Pept Lett. 2009;16(4):454-6. PMID:19356147
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 Qian H, Zhao X, Yan R, Yao X, Gao S, Sun X, Du X, Yang H, Wong CCL, Yan N. Structural basis for catalysis and substrate specificity of human ACAT1. Nature. 2020 May;581(7808):333-338. doi: 10.1038/s41586-020-2290-0. Epub 2020 May, 13. PMID:32433614 doi:http://dx.doi.org/10.1038/s41586-020-2290-0
- ↑ 4.0 4.1 Cases S, Novak S, Zheng YW, Myers HM, Lear SR, Sande E, Welch CB, Lusis AJ, Spencer TA, Krause BR, Erickson SK, Farese RV Jr. ACAT-2, a second mammalian acyl-CoA:cholesterol acyltransferase. Its cloning, expression, and characterization. J Biol Chem. 1998 Oct 9;273(41):26755-64. doi: 10.1074/jbc.273.41.26755. PMID:9756919 doi:http://dx.doi.org/10.1074/jbc.273.41.26755
- ↑ 5.0 5.1 Guan C, Niu Y, Chen SC, Kang Y, Wu JX, Nishi K, Chang CCY, Chang TY, Luo T, Chen L. Structural insights into the inhibition mechanism of human sterol O-acyltransferase 1 by a competitive inhibitor. Nat Commun. 2020 May 18;11(1):2478. doi: 10.1038/s41467-020-16288-4. PMID:32424158 doi:http://dx.doi.org/10.1038/s41467-020-16288-4
- ↑ Liu J, Chang CC, Westover EJ, Covey DF, Chang TY. Investigating the allosterism of acyl-CoA:cholesterol acyltransferase (ACAT) by using various sterols: in vitro and intact cell studies. Biochem J. 2005 Oct 15;391(Pt 2):389-97. doi: 10.1042/BJ20050428. PMID:15992359 doi:http://dx.doi.org/10.1042/BJ20050428
- ↑ Rogers MA, Liu J, Song BL, Li BL, Chang CC, Chang TY. Acyl-CoA:cholesterol acyltransferases (ACATs/SOATs): Enzymes with multiple sterols as substrates and as activators. J Steroid Biochem Mol Biol. 2015 Jul;151:102-7. doi: 10.1016/j.jsbmb.2014.09.008., Epub 2014 Sep 12. PMID:25218443 doi:http://dx.doi.org/10.1016/j.jsbmb.2014.09.008
- ↑ Hartmann T, Kuchenbecker J, Grimm MO. Alzheimer's disease: the lipid connection. J Neurochem. 2007 Nov;103 Suppl 1:159-70. doi: 10.1111/j.1471-4159.2007.04715.x. PMID:17986151 doi:http://dx.doi.org/10.1111/j.1471-4159.2007.04715.x
- ↑ Li J, Gu D, Lee SS, Song B, Bandyopadhyay S, Chen S, Konieczny SF, Ratliff TL, Liu X, Xie J, Cheng JX. Abrogating cholesterol esterification suppresses growth and metastasis of pancreatic cancer. Oncogene. 2016 Dec 15;35(50):6378-6388. doi: 10.1038/onc.2016.168. Epub 2016 May , 2. PMID:27132508 doi:http://dx.doi.org/10.1038/onc.2016.168
- ↑ Rudel LL, Shelness GS. Cholesterol esters and atherosclerosis-a game of ACAT and mouse. Nat Med. 2000 Dec;6(12):1313-4. doi: 10.1038/82110. PMID:11100106 doi:http://dx.doi.org/10.1038/82110
Student Contributors
- Leah Goehring
- Gabby Smith
- Anna Campbell