This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Replication Termination Protein
From Proteopedia
(Difference between revisions)
| (3 intermediate revisions not shown.) | |||
| Line 2: | Line 2: | ||
== Function == | == Function == | ||
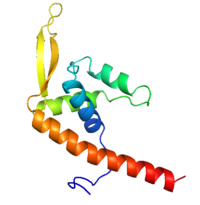
[[Image:RTP.png|200px|left|thumb| Diagram of RTP monomer with secondary structure highlighted.]] | [[Image:RTP.png|200px|left|thumb| Diagram of RTP monomer with secondary structure highlighted.]] | ||
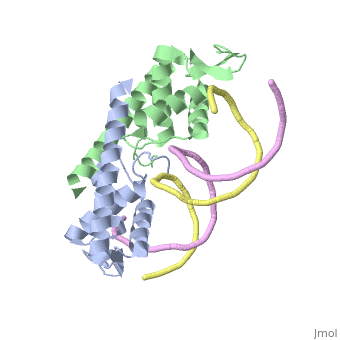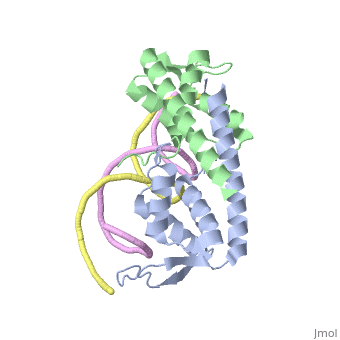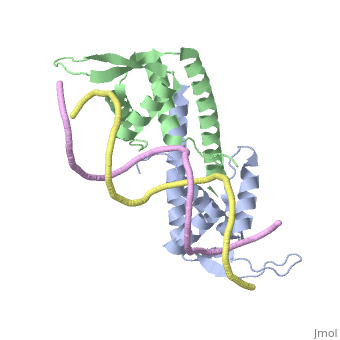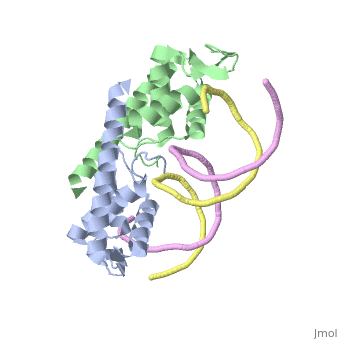
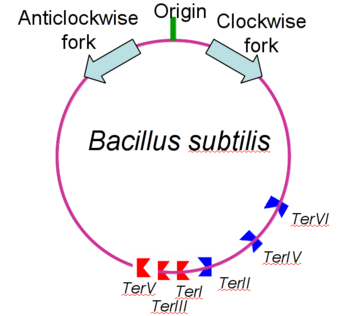
| - | The '''replication termination protein''' or '''replication terminator protein''' (RTP) is one of only two well-defined proteins known to be involved in arresting DNA replication forks, the other being a protein known as '''Tus''' ('''termination utilisation substance''' or '''DNA replication terminus site-binding protein''') from ''E. coli'' <ref> Kamada K, Horiuchi T, Ohsumi K, Shimamoto N, Morkikawa K, (1996) Structure of a replication-terminator protein complexed with DNA. Nature, 383:598-603 </ref>. RTP was discovered in ''Bacillus subtilis'' and has been identified as a DNA binding protein of the winged helix family that forms a dimer of 29kDa. This dimeric form has been shown to have an exceptionally high affinity for its cognate binding sites( Kd ~10-11M-1)<ref> Wilce et. al. (2001) Structure of the RTP−DNA complex and the mechanism of polar replication fork arrest. Nature Structural Biology, 8:206-210 </ref>, otherwise known as Termination sites (Ter sites). These Ter sites are found in multiple locations in the ''B. subtilis'' genome <ref> Gautam A. et.al. (2001) A single domain of the replication termination protein of ''Bacillus subtilis'' is involved in arresting both DnaB helicase and RNA polymerase. Journal of Biological Chemistry, 276:23471-23479</ref>. For more details see:<br /> | + | The '''replication termination protein''' or '''replication terminator protein''' (RTP) is one of only two well-defined proteins known to be involved in arresting DNA replication forks, the other being a protein known as '''Tus''' ('''termination utilisation substance''' or '''DNA replication terminus site-binding protein''' or '''ter-binding protein)''' from ''E. coli'' <ref> Kamada K, Horiuchi T, Ohsumi K, Shimamoto N, Morkikawa K, (1996) Structure of a replication-terminator protein complexed with DNA. Nature, 383:598-603 </ref>. RTP was discovered in ''Bacillus subtilis'' and has been identified as a DNA binding protein of the winged helix family that forms a dimer of 29kDa. This dimeric form has been shown to have an exceptionally high affinity for its cognate binding sites( Kd ~10-11M-1)<ref> Wilce et. al. (2001) Structure of the RTP−DNA complex and the mechanism of polar replication fork arrest. Nature Structural Biology, 8:206-210 </ref>, otherwise known as Termination sites (Ter sites). These Ter sites are found in multiple locations in the ''B. subtilis'' genome <ref> Gautam A. et.al. (2001) A single domain of the replication termination protein of ''Bacillus subtilis'' is involved in arresting both DnaB helicase and RNA polymerase. Journal of Biological Chemistry, 276:23471-23479</ref>. For more details see:<br /> |
*[[RTP and Tus]]<br /> | *[[RTP and Tus]]<br /> | ||
*[[Rtp and Tus DNA Binding]]<br /> | *[[Rtp and Tus DNA Binding]]<br /> | ||
| Line 119: | Line 119: | ||
Debate over the mechanism of Replication termination by RTP has led to a number of mechanisms being proposed. Original work on this question was hampered by an incorrect structure of RTP being released. This structure was produced in solution and was shown to be a symmetric dimer<ref> Bastia D. (1995) Crystal structure of the replication terminator protein from b. subtilis at 2.6 A. Cell 80: 651-660 </ref> Later crystallisation of the protein bound to DNA showed the RTP formed a asymmetric dimer when bound to cognate sequences <ref> Wilce et. al. (2001) Structure of the RTP−DNA complex and the mechanism of polar replication fork arrest. Nature Structural Biology, 8:206-210</ref> . One early study proposed that the asymmetric interaction between terminator protein and terminator DNA contributed to the observed polarity, with later studies showing that the RTP/Ter complex contained partially unwound DNA, suggesting a locked complex was responsible. More recent data has implicated that protein -protein interactions between RTP and the helicase is primarily responsible for termination and that this mechanism is modulated by the asymmetrical interactions described above <ref>Kaplan D.L., Bastia D. (2009). Mechanisms of polar arrest of a replication fork. Molecular Microbiology 72: 279-284</ref>. | Debate over the mechanism of Replication termination by RTP has led to a number of mechanisms being proposed. Original work on this question was hampered by an incorrect structure of RTP being released. This structure was produced in solution and was shown to be a symmetric dimer<ref> Bastia D. (1995) Crystal structure of the replication terminator protein from b. subtilis at 2.6 A. Cell 80: 651-660 </ref> Later crystallisation of the protein bound to DNA showed the RTP formed a asymmetric dimer when bound to cognate sequences <ref> Wilce et. al. (2001) Structure of the RTP−DNA complex and the mechanism of polar replication fork arrest. Nature Structural Biology, 8:206-210</ref> . One early study proposed that the asymmetric interaction between terminator protein and terminator DNA contributed to the observed polarity, with later studies showing that the RTP/Ter complex contained partially unwound DNA, suggesting a locked complex was responsible. More recent data has implicated that protein -protein interactions between RTP and the helicase is primarily responsible for termination and that this mechanism is modulated by the asymmetrical interactions described above <ref>Kaplan D.L., Bastia D. (2009). Mechanisms of polar arrest of a replication fork. Molecular Microbiology 72: 279-284</ref>. | ||
| - | + | ||
==3D structures of replication termination protein== | ==3D structures of replication termination protein== | ||
Updated on {{REVISIONDAY2}}-{{MONTHNAME|{{REVISIONMONTH}}}}-{{REVISIONYEAR}} | Updated on {{REVISIONDAY2}}-{{MONTHNAME|{{REVISIONMONTH}}}}-{{REVISIONYEAR}} | ||
| + | {{#tree:id=OrganizedByTopic|openlevels=0| | ||
| - | [[1bm9]] – BsRTP – ''Bacillus subtilis''<br /> | + | *RTP |
| - | [[1j0r]], [[2dqr]] - BsRTP (mutant)<br /> | + | |
| - | [[2dpd]] - BsRTP + DNA<br /> | + | **[[1bm9]] – BsRTP – ''Bacillus subtilis''<br /> |
| - | [[1f4k]], [[2dpu]], [[2efw]] – BsRTP (mutant) + DNA<br /> | + | **[[1j0r]], [[2dqr]] - BsRTP (mutant) <br /> |
| - | [[1ecr]] – | + | **[[2dpd]] - BsRTP + DNA<br /> |
| + | **[[1f4k]], [[2dpu]], [[2efw]] – BsRTP (mutant) + DNA<br /> | ||
| + | |||
| + | *Tus | ||
| + | |||
| + | **[[1ecr]], [[2ewj]], [[2i05]], [[2i06]], [[4xr0]], [[4xr1]], [[4xr3]] – EcTus + DNA – ''Escherichia coli''<br /> | ||
| + | **[[4xr2]] – EcTus (mutant) + DNA <br /> | ||
| + | }} | ||
==References== | ==References== | ||
{{reflist}} | {{reflist}} | ||
| - | + | </StructureSection> | |
[[Category:Topic Page]] | [[Category:Topic Page]] | ||
Current revision
| |||||||||||
Proteopedia Page Contributors and Editors (what is this?)
Michal Harel, Alexander Berchansky, Craig Mooney, Joel L. Sussman