We apologize for Proteopedia being slow to respond. For the past two years, a new implementation of Proteopedia has been being built. Soon, it will replace this 18-year old system. All existing content will be moved to the new system at a date that will be announced here.
User:Marcos Ngo/Sandbox 1
From Proteopedia
< User:Marcos Ngo(Difference between revisions)
| (154 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
| - | + | =='''Human NTHL1'''== | |
| - | <StructureSection load='7rds' size='340' | + | <StructureSection load='7rds' size='340' |
| + | caption='Human NTHL1 at 2.5 Å [[resolution]]. The yellow and brown ligand is the iron-sulfur cluster.' | ||
| + | scene=''> | ||
| + | ==Background and Function== | ||
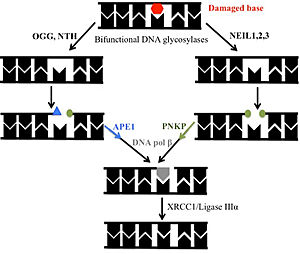
| + | [[DNA glycosylases]] search the entire genome for [[DNA]] lesions. These highly selective enzymes recognize a damaged base and remove it. There are four super families of glycosylases: Udg, Nth, Nei, and AGG. '''hNTHL1''' or '''human Endonuclease III''' (Nth family) is a 34 kDa bifunctional DNA glycosylase belonging to the HhH (Helix-Hairpin-Helix) superfamily. It is involved in the base excision repair (BER) process. A bifunctional glycosylase refers to the ability to '''excise''' damaged bases and '''nick''' the backbone. hNTHL1 preferentially excises oxidized pyrimidines. Thymine glycol (Tg) is the preferred substrate. Upon encountering this damaged base, the protein severs the N-glycosidic bond, leaving an apurinic site. From here, the backbone is cleaved via beta elimination, which leaves a 3’ aldehyde and creates a single-strand break. Next, the DNA is handed off to [[Apurinic-Apyrimidinic Endonuclease]] (APE1) or [https://proteopedia.org/wiki/index.php/1yj5 Polynucleotide Kinase], leaving a free 3′ hydroxyl for [[DNA polymerase beta]] to insert the correct nucleotide. Finally, the nick is sealed by [https://proteopedia.org/wiki/index.php/DNA_ligase DNA ligase IIIα]<ref>PMID:34871433</ref><ref>PMID:20005182</ref><ref>PMID:9295348</ref>. | ||
| - | + | [[Image:DNAGly.jpg |300px|]] | |
| - | [ | + | |
| - | + | The gene encoding hNTHL1 is NTHL1, which is located on chromosome 16. It is widely expressed across tissues, with the highest levels observed in the heart. This elevated expression may reflect the heart’s high demand for ATP production, which generates significant oxidative stress and thus increases reliance on base excision repair (BER) proteins for genome maintenance. Additionally, hNTHL1 expression is regulated during the cell cycle, with [[transcription]] levels rising during the early and mid S phases <ref>https://www.uniprot.org/uniprotkb/P78549/entry</ref><ref>PMID:8990169</ref><ref>PMID:10882850</ref>. | |
| - | + | ||
| - | + | ||
| - | + | hNTHL1 has been observed in both the nucleus and mitochondria, meaning that the protein has dual transport signals to repair damaged bases. Nuclear localization signals (NLS) and mitochondrial localization signal (MLS) have been observed around the N-terminal region <ref>https://www.uniprot.org/uniprotkb/P78549/entry</ref><ref>PMID:10882850</ref><ref>PMID:9705289</ref><ref>PMID:1478671</ref>. | |
| - | + | == Mechanism of Repair == | |
| + | DNA glycosylases remove damaged bases through a “pinch, push, plug, and pull” mechanism. First, the DNA is “pinched” by the enzyme, which destabilizes the helix. Next, they use a wedge amino acid to “push” the lesion out of the helix. While the lesion is being flipped out, another amino acid “plugs” into the helix to fill the gap and maintain the structure of the helix. Finally, the lesion is “pulled” into the active site to allow for lesion removal <ref>https://scholarworks.uvm.edu/cgi/viewcontent.cgi?article=2160&context=graddis</ref><ref>PMID:20469926</ref><ref>PMID:12220189</ref>. | ||
| + | Recent studies examined how hNTHL1 initiates BER of oxidative lesions in nucleosomal DNA. When lesions are positioned away from the [[histone]] core, hNTHL1 processes them with nearly the same efficiency as in naked DNA (without histones). When lesions faced inward toward the histone core, repair was initially poor but improved significantly at higher hNTHL1 concentrations. Additionally, sections near the edge of the nucleosome were repaired more efficiently than those near the nucleosome center. This suggests that both the partial unwrapping of DNA from the histone core and the positioning of the lesion outward from the nucleosome edge allow hNTHL1 to efficiently access and repair DNA lesions <ref>PMID:17923696</ref>. | ||
| - | < | + | == Structural Highlights == |
| + | <table><tr><td colspan='2'> | ||
</td></tr><tr id='method'><td class="sblockLbl"><b>[[Empirical_models|Method:]]</b></td><td class="sblockDat" id="methodDat">X-ray diffraction, [[Resolution|Resolution]] 2.5Å</td></tr> | </td></tr><tr id='method'><td class="sblockLbl"><b>[[Empirical_models|Method:]]</b></td><td class="sblockDat" id="methodDat">X-ray diffraction, [[Resolution|Resolution]] 2.5Å</td></tr> | ||
<tr id='ligand'><td class="sblockLbl"><b>[[Ligand|Ligands:]]</b></td><td class="sblockDat" id="ligandDat"><scene name='pdbligand=SF4:IRON/SULFUR+CLUSTER'>SF4</scene></td></tr> | <tr id='ligand'><td class="sblockLbl"><b>[[Ligand|Ligands:]]</b></td><td class="sblockDat" id="ligandDat"><scene name='pdbligand=SF4:IRON/SULFUR+CLUSTER'>SF4</scene></td></tr> | ||
| Line 21: | Line 25: | ||
</table> | </table> | ||
| - | = | + | hNTHL1 consists of <scene name='10/1077482/Two_domains/2'>two alpha-helical domains</scene> connected by two linkers. <scene name='10/1077482/Ncfedomain1/5'>Domain 1</scene> has the iron sulfur cluster, N- and C-termini, and a catalytic residue ('''Asp 239'''). <scene name='10/1077482/Domain2features/3'>Domain 2</scene> has six helical barrels, helix-hairpin-helix, and the other catalytic residue ('''Lys 220'''). The <scene name='10/1077482/Proglyhhh/1'>HhH</scene> motif has a characteristic glycine and proline-rich loop. The HhH allows for hydrogen bond interactions with the DNA backbone <ref>PMID:12840008</ref><ref>https://scholarworks.uvm.edu/cgi/viewcontent.cgi?article=2160&context=graddis</ref><ref>PMID:1283262</ref>. |
| - | + | ||
| - | ''' | + | This structure is captured in an <scene name='10/1077482/Open_conformation/1'>open conformation</scene> where the catalytic residues Lys220 and Asp239 are positioned approximately 25 Å apart, which is too far for catalysis. This implies that a conformational change is required to assemble the active site. To find the closed conformation, an engineered chimera was made by swapping the <scene name='10/1077482/Linker1/1'>flexible interdomain linker</scene> in human NTHL1 with a shorter, more rigid linker from a bacterial homolog. The [https://proteopedia.org/wiki/index.php/7rdt hNTHL1 chimera] structure adopts a <scene name='10/1077482/Chimera/1'>closed conformation</scene> where Lys220 and Asp239 are approximately 5 Å apart, which mimics the configuration seen in catalytically active homologs. The linker is not fully modeled due to disorder in the electron density map <ref>PMID:34871433</ref>. |
| - | ''' | + | The role of the <scene name='10/1077482/Fes_proper/4'>FeS Cluster</scene> is highly debated. One of the views is that the cluster is involved in scanning for lesions. Researchers found that oxidizing the FeS cluster in hNTHL1 from [4Fe-4S]^2+ to [4Fe-4S]^3+ increases its binding to DNA. When a mismatch such as C:A is introduced, this can disrupt DNA charge transport not allowing electrons to travel along the helix. This could stop the reduction of [4Fe-4S]^3+ to [4Fe-4S]^2+, leaving Nth bound until all lesions are removed. Another view is that the FeS cluster plays a role as a structural scaffold to stabilize the interaction of the protein with the DNA. A <scene name='10/1077482/Cys_and_fes/1'>Cys-Xaa6-Cys-Xaa2-Cys-Xaa5-Cys</scene> motif binds the iron sulfur cluster <ref>PMID:19720997</ref><ref>PMID:28817778</ref><ref>DOI:https://pubs.rsc.org/en/content/articlelanding/2022/cc/d2cc03643f</ref><ref>PMID:8990169</ref>. |
| + | |||
| + | The function of the <scene name='10/1077482/N-terminus_AlphaFold/3'>N-terminus</scene> (AlphaFold Prediction) of hNTHL1 has been a subject of study <ref>PMID:34293799</ref><ref>PMID:37933859</ref>. It is theorized that the N-terminus, which is extended compared to homologs, functions as a means to remain bound to DNA, protecting the labile abasic site while waiting for its handoff with APE1. This was found through truncation of the N-terminal region (residues 1-96), which revealed that deletion of 55, 75, or 80 residues from the N-terminus resulted in a four to fivefold increase in catalytic activity. The rate-limiting step in hNTHL1's reaction is the release of the free 3’ aldehyde <ref>PMID:12144783</ref>. | ||
| + | |||
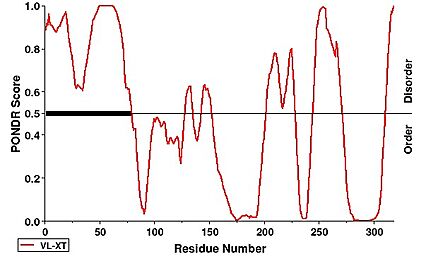
| + | Notably, the first 63 residues of the N-terminus were not modeled due to disorder. This can be observed under a PONDR prediction | ||
| + | [[Image:PONDR.jpg|438 × 271px|'''PONDR Disorder Prediction''']] <ref>https://www.pondr.com/</ref>. | ||
| + | |||
| + | Additionally, several motifs are present within the extended N-terminus of hNTHL1. A bipartite nuclear localization signal (NLS) spans residues 28–52, while an overlapping region of basic and acidic residues ranges from residues 23–42. Two additional NLS motifs, characterized by clusters of basic residues, are located at residues 48–56 and 90–100. A mitochondrial localization signal (MLS) is also predicted at residues 1–22 <ref>https://www.uniprot.org/uniprotkb/P78549/entry</ref><ref>PMID:10882850</ref><ref>PMID:1478671</ref>. | ||
| + | |||
| + | == Disease == | ||
| + | When the base excision repair pathway is compromised, the ability to repair damaged DNA is significantly reduced. This causes mutations throughout the genome, leading to the progression of cancer. '''NTHL1-Tumor Syndrome''' is a disease caused by variants affecting the gene that render the glycosylase inactive. This syndrome is diagnosed by a germline biallelic pathogenic variant, which can be found through genetic testing. Upon diagnosis, one's cumulative lifetime risk of developing extracolonic cancer by age 60 is 35-78%. This syndrome is characterized by an increased risk of colorectal cancer, breast cancer, and colorectal polyposis. Around 5% of colorectal cancers can be explained by germline mutations within a CRC predisposing gene. Exome sequencing has led to the identification of a homozygous nonsense mutation (c.268C>T encoding p.Q90*) in the base excision repair gene NTHL1 in three unrelated families <ref>PMID:32239880</ref><ref>PMID:25938944</ref><ref>https://www.uniprot.org/uniprot/NTH_HUMAN NTH_HUMAN</ref>. | ||
| - | + | A functional, non-mutated hNTHL1 can also contribute to cancer cell survival. In triple-negative breast cancer, the protein [https://proteopedia.org/wiki/index.php/9b4p BCL11A] is frequently overexpressed. BCL11A is a transcription factor shown to stimulate hNTHL1 activity, enhancing base excision repair (BER) and enabling cancer cells to proliferate in high levels of oxidative damage. Separately, [https://proteopedia.org/wiki/index.php/5ytx Y-box binding protein-1] (YB-1) is overexpressed in tumor cells, and hNTHL1 can be activated through direct interaction with YB-1. This boosts its ability to process oxidized bases. This YB–1–mediated stimulation of hNTHL1 causes resistance to cisplatin, a form of chemotherapy, allowing for cancer proliferation <ref>PMID:36186110</ref><ref>PMID:18307537</ref>. | |
== References == | == References == | ||
<references/> | <references/> | ||
| - | __TOC__ | ||
| - | </StructureSection> | ||
[[Category: Homo sapiens]] | [[Category: Homo sapiens]] | ||
[[Category: Large Structures]] | [[Category: Large Structures]] | ||
Current revision
Human NTHL1
| |||||||||||