This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox caroldavila
From Proteopedia
| Line 1: | Line 1: | ||
| - | + | {{STRUCTURE_1hzh| right| PDB=1hzh | SCENE=Antibody/1hzh_starting_scene/3 |CAPTION= Structura IGG B12 uman: o mostra a unui posibil vaccin pentru HIV, [[1hzh]] }} | |
| - | {{STRUCTURE_1hzh| right| PDB=1hzh | SCENE=Antibody/1hzh_starting_scene/3 |CAPTION= | + | |
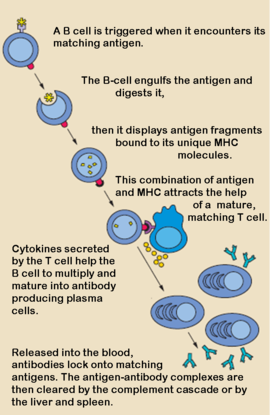
'''Antibodies''', also known as Immunoglobulins (Ig) are gamma globulin proteins, primarily found in the blood of vertebrates. These [[glycoproteins]] serve as a critical component of the immune system when the host fails to activate alternative compliment pathways or phagocytic cells in response to invading microorganisms or other [http://en.wikipedia.org/wiki/Antigen antigens]. The incredible specificity with which immunoglobulins bind to an antigen is based upon structural complementarity between the antigen and antibody <scene name='Antibody/1hzh_heavy_chains/1'>heavy </scene>and <scene name='Antibody/1hzh_light_chains/1'>light chains </scene>. It is this specificity that has made <scene name='Antibody/1hzh_starting_scene/3'>antibodies</scene> a critical component in laboratory and medical research. | '''Antibodies''', also known as Immunoglobulins (Ig) are gamma globulin proteins, primarily found in the blood of vertebrates. These [[glycoproteins]] serve as a critical component of the immune system when the host fails to activate alternative compliment pathways or phagocytic cells in response to invading microorganisms or other [http://en.wikipedia.org/wiki/Antigen antigens]. The incredible specificity with which immunoglobulins bind to an antigen is based upon structural complementarity between the antigen and antibody <scene name='Antibody/1hzh_heavy_chains/1'>heavy </scene>and <scene name='Antibody/1hzh_light_chains/1'>light chains </scene>. It is this specificity that has made <scene name='Antibody/1hzh_starting_scene/3'>antibodies</scene> a critical component in laboratory and medical research. | ||
Revision as of 07:17, 20 October 2010
| |||||||||
| Structura IGG B12 uman: o mostra a unui posibil vaccin pentru HIV, 1hzh | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Ligands: | , , , , | ||||||||
| |||||||||
| |||||||||
| Resources: | FirstGlance, OCA, RCSB, PDBsum | ||||||||
| Coordinates: | save as pdb, mmCIF, xml | ||||||||
Antibodies, also known as Immunoglobulins (Ig) are gamma globulin proteins, primarily found in the blood of vertebrates. These glycoproteins serve as a critical component of the immune system when the host fails to activate alternative compliment pathways or phagocytic cells in response to invading microorganisms or other antigens. The incredible specificity with which immunoglobulins bind to an antigen is based upon structural complementarity between the antigen and antibody and . It is this specificity that has made a critical component in laboratory and medical research.