This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Copper Amine Oxidase
From Proteopedia
(Difference between revisions)
| Line 36: | Line 36: | ||
== Reaction == | == Reaction == | ||
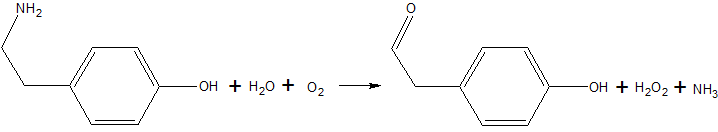
| - | Copper amine oxidase catalyzes the oxidation of a primary amine to | + | Copper amine oxidase catalyzes the oxidation of a primary amine to the corresponding aldehyde, yielding hydrogen peroxide and free ammonia. An example of this is the oxidation of [http://en.wikipedia.org/wiki/Tyramine tyramine]: |
[[Image:Tyramine oxidation.png|The oxidation of tyramine.]] | [[Image:Tyramine oxidation.png|The oxidation of tyramine.]] | ||
Revision as of 06:08, 15 March 2011
| This Sandbox is Reserved from January 10, 2010, through April 10, 2011 for use in BCMB 307-Proteins course taught by Andrea Gorrell at the University of Northern British Columbia, Prince George, BC, Canada. |
To get started:
More help: Help:Editing |
2d1w is a copper amine oxidase found in Arthrobacter globiformis.
Contents |
Structure
Ligand
A closeup of the Cu2+ .
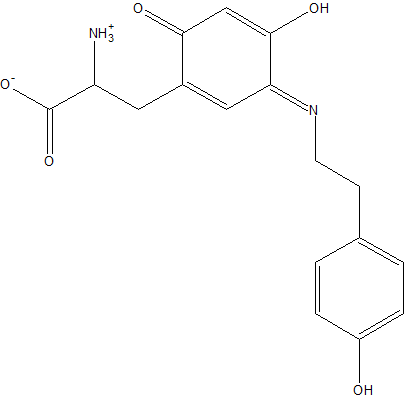
Modified Residue
Reaction
Copper amine oxidase catalyzes the oxidation of a primary amine to the corresponding aldehyde, yielding hydrogen peroxide and free ammonia. An example of this is the oxidation of tyramine:
References
Proteopedia Page Contributors and Editors (what is this?)
Michal Harel, Raymond Lyle, Alexander Berchansky, OCA, Jaime Prilusky