This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Momodou L. Jammeh/Sandbox 1
From Proteopedia
(→STRUCTURE) |
|||
| Line 10: | Line 10: | ||
===STRUCTURE=== | ===STRUCTURE=== | ||
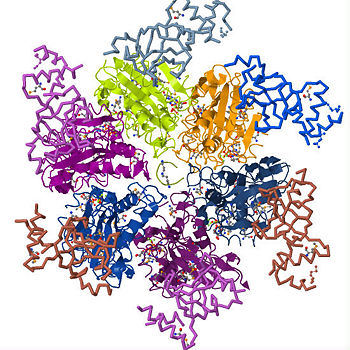
| - | <Structure load='3ICE' size='500' frame='true' align='right' caption='Rho factor in its hexameric form with its six identical subunits forming a helicase and an RNA molecule right through the center' /> Rho factor is a hexamer of six identical subunits that interact to form a closed, dynamic structure called a <scene name='User:Momodou_L._Jammeh/Sandbox_1/Hexameric_rho/2'>helicase.</scene> It is made up of a single polypeptide chain of about 419 amino acid residues. Each subunit has an <scene name='User:Momodou_L._Jammeh/Sandbox_1/Hexameric_rho_n-terminal/2'>amino-terminus</scene> (residues in blue and light blue) and <scene name='User:Momodou_L._Jammeh/Sandbox_1/Hexameric_rho_c-terminal/1'>carboxyl-terminus</scene> (residues in red and orange) domains with the former on the periphery of the subunits and the latter, which is also the main catalytic site on the central, upper portion of the hexamer. The region connecting each of the subunits to for the closed ring structure serves as the <scene name='User:Momodou_L._Jammeh/Sandbox_1/Subunits_with_adp_molecules/5'>ATPase</scene> site for the generation of energy for rho function and the <scene name='User:Momodou_L._Jammeh/Sandbox_1/Subunits_with_adp_molecules/4'>RNA</scene> occupying the center of rho. | + | <Structure load='3ICE' size='500' frame='true' align='right' caption='Rho factor in its hexameric form with its six identical subunits forming a helicase and an RNA molecule right through the center' /> Rho factor is a hexamer of six identical subunits that interact to form a closed, dynamic structure called a <scene name='User:Momodou_L._Jammeh/Sandbox_1/Hexameric_rho/2'>helicase.</scene> It is made up of a single polypeptide chain of about 419 amino acid residues which comprise the alpha-helix and <scene name='User:Momodou_L._Jammeh/Sandbox_1/Rho_ball_sheets/3'>beta sheet</scene> conformations that make up the secondary structure of each subunit. Each subunit has an <scene name='User:Momodou_L._Jammeh/Sandbox_1/Hexameric_rho_n-terminal/2'>amino-terminus</scene> (residues in blue and light blue) and <scene name='User:Momodou_L._Jammeh/Sandbox_1/Hexameric_rho_c-terminal/1'>carboxyl-terminus</scene> (residues in red and orange) domains with the former on the periphery of the subunits and the latter, which is also the main catalytic site on the central, upper portion of the hexamer. The region connecting each of the subunits to for the closed ring structure serves as the <scene name='User:Momodou_L._Jammeh/Sandbox_1/Subunits_with_adp_molecules/5'>ATPase</scene> site for the generation of energy for rho function and the <scene name='User:Momodou_L._Jammeh/Sandbox_1/Subunits_with_adp_molecules/4'>RNA</scene> occupying the center of rho. |
---- | ---- | ||
===INHIBITION=== | ===INHIBITION=== | ||
---- | ---- | ||
Revision as of 21:18, 24 April 2011
Contents |
RHO TERMINATION FACTOR
INTRODUCTION
Transcription is the process of mRNA synthesis by RNA polymerase, an enzyme that uses a strand of DNA as a template for ribonucloetide addition. During this process, RNA polymerase encounters several regulatory proteins, called transcription factors, that effect transcription in various ways. One of the major regulatory activities of transcription regulators is terminating the process at specific sites in the transcription unit. In E. coli and prokaryotes in general, transcription is terminated through rho dependent and independent mechanisms. Transcription termination without rho occurs through RNA polymerase's intrinsic ability to terminate transcription in response to certain, limited sequences in the template DNA strand. Rho dependent termination requires a helicase protein called rho factor which belongs in the helicase class of enzymes. Helicases are motor proteins that function through directional movement on the nucleic acid phosphodiester backbone and can dissociate bound DNA-DNA, RNA-DNA or RNA-RNA strands. Rho specifically terminates transcription by separating the the new RNA strand annealed to the DNA strand being transcribed. As pictured on the upper right corner, it is a hexameric ring-shaped helicase that uses energy derived from its ATPase mechanism (hydrolysis of ATP into ADP and an organic phosphate) to drive its movement along the newly formed RNA molecule toward the elongation complex to be dissociated. While rho is only requirement for the termination process, additional elongation factors such as NusA and NusG have been shown to enhance the process. NusA reduces the elongation rate allowing rho to catch up with RNA polymerase despite its high processivity while NusG enhances the rate of RNA release by coupling rho to the elongation complex. Since its discovery in 1969, studies on rho have been essential in understanding how external factors regulation transcription termination and its structure provides insight into how several stable hexameric helicases load onto their nucleic acid substrates.
STRUCTURE
|