This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox 39
From Proteopedia
(Difference between revisions)
| Line 2: | Line 2: | ||
{{Template: Oberholser Sandbox Reservation}} | {{Template: Oberholser Sandbox Reservation}} | ||
| - | <StructureSection load='9pap' size=' | + | <StructureSection load='9pap' size='300' side='right' caption='Structure of Papain(PDB entry [[9pap]])' scene=''> |
=='''Papain'''== | =='''Papain'''== | ||
| Line 10: | Line 10: | ||
== Function == | == Function == | ||
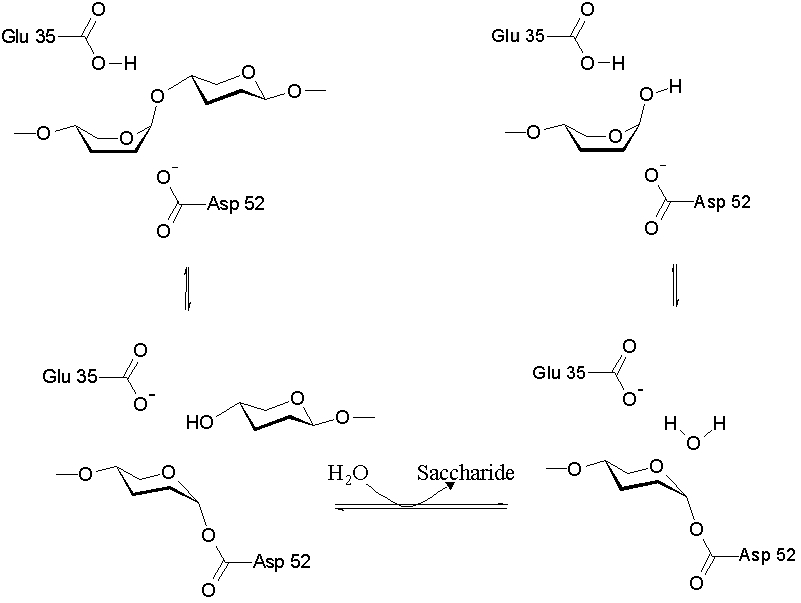
| - | As a cysteine protease, Papain utilizes a nucleophilic cysteine thiol as part of its catalytic triad. | + | As a cysteine protease, Papain utilizes a nucleophilic cysteine thiol as part of its catalytic triad. Papain's Cys-25 is deprotonated by its His-159. The now nucleophilic Cys-25 attacks the carbonyl carbon of the peptide backbone, forming an acyl enzyme intermediate in which the peptide's amino terminal is free. Also in this step, His-159 is returned to its deprotonated form. The intermediate is then deacylated by a water molecule, and it releases the carboxyl terminal of the peptide to produce the product and regenerate the active enzyme. This entire mechanism is shown below: |
[[Image:jrip.jpg]] | [[Image:jrip.jpg]] | ||
Revision as of 23:14, 8 November 2011
| Please do NOT make changes to this Sandbox. Sandboxes 30-60 are reserved for use by Biochemistry 410 & 412 at Messiah College taught by Dr. Hannah Tims during Fall 2012 and Spring 2013. |
| |||||||||||