This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox 50
From Proteopedia
(Difference between revisions)
| Line 4: | Line 4: | ||
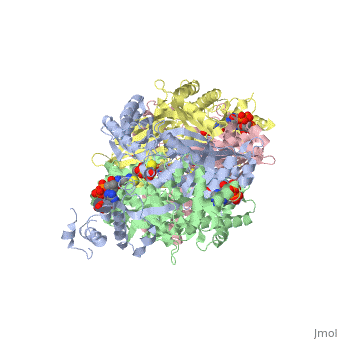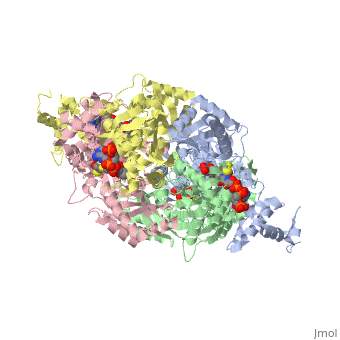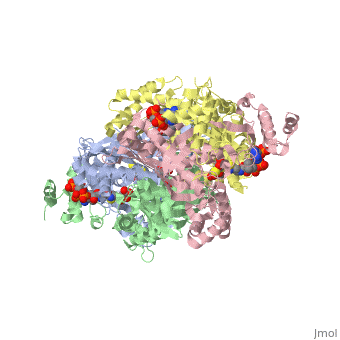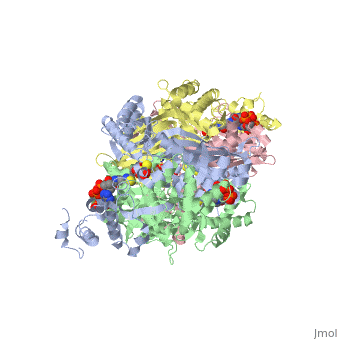
= Horse Pancreatic Lipase = | = Horse Pancreatic Lipase = | ||
Lipase, which is produced primarily in the pancreas, functions as a catalyst in the hydrolysis of ester bonds in lipid substrates. This makes lipases an essential molecule for fat digestion. | Lipase, which is produced primarily in the pancreas, functions as a catalyst in the hydrolysis of ester bonds in lipid substrates. This makes lipases an essential molecule for fat digestion. | ||
| - | Horse pancreatic lipase’s function is to convert triacylglycerols into 2-monoacylglycerols and free fatty acids. These monomers are then able to be shuttled into the small intestine to be absorbed into the lymphatic system. | + | Horse pancreatic lipase’s (HPL)function is to convert triacylglycerols into 2-monoacylglycerols and free fatty acids. These monomers are then able to be shuttled into the small intestine to be absorbed into the lymphatic system. |
==Structure == | ==Structure == | ||
| - | Horse pancreatic lipase contains two identical peptide chains containing 449 amino acid residues. The N to C terminal order is shown <scene name='Sandbox_50/5-3_rainbow/1'>here</scene> where the N terminus is in blue and can be followed to the C terminus in red. | + | Horse pancreatic lipase contains two identical peptide chains containing 449 amino acid residues. The N to C terminal order is shown <scene name='Sandbox_50/5-3_rainbow/1'>here</scene> where the N terminus is in blue and can be followed to the C terminus in red. Each chain contains |
| + | <scene name='Sandbox_50/N_and_c_terminus/1'>two well defined domains</scene>. The N-terminal domain is shown in blue and the C terminal domain is shown in green. The active site of HPL is highlighted in red to show its location in the N terminal domain of the A chain. | ||
=== test === | === test === | ||
| + | |||
| + | <scene name='Sandbox_50/N_and_c_terminus/1'>TextToBeDisplayed</scene> | ||
<scene name='Sandbox_50/Hphobic_residues/2'>h phobic facing in in gray</scene> | <scene name='Sandbox_50/Hphobic_residues/2'>h phobic facing in in gray</scene> | ||
Revision as of 21:12, 13 November 2011
| Please do NOT make changes to this Sandbox. Sandboxes 30-60 are reserved for use by Biochemistry 410 & 412 at Messiah College taught by Dr. Hannah Tims during Fall 2012 and Spring 2013. |
| |||||||||||