This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Group:MUZIC:FilaminC
From Proteopedia

| Line 1: | Line 1: | ||
| - | {{STRUCTURE_1v05| PDB=1v05 | SIZE=300| SCENE= ''User:Ritika_Sethi/workbench/FilaminC/Flnc_ig_24/1'|right|CAPTION=Human Filamin C domain 24}} | ||
| - | |||
| - | <scene name='User:Ritika_Sethi/workbench/FilaminC/Flnc_ig_24/1'>Human Filamin C domain 24</scene> | ||
| - | |||
| - | |||
| - | |||
{{<StructureSection load='1v05' size='500' side='left' scene='User:Ritika_Sethi/workbench/FilaminC/Flnc_ig_24/1' caption='Human Filamin C domain 24'>}} | {{<StructureSection load='1v05' size='500' side='left' scene='User:Ritika_Sethi/workbench/FilaminC/Flnc_ig_24/1' caption='Human Filamin C domain 24'>}} | ||
Revision as of 09:09, 29 November 2011
{{
}}
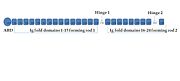
Filamin CIn humans, 3 isoforms of Filamins exist that are coded by 3 different genes. While the genes for Filamin A and Filamin B are present on the X chromosome and chromosome 3 respectively, and both show a ubiquitous expression in many tissues, gene for Filamin C is located on the Chromosome 7 and the encoded protein is specifically expressed in muscles and has been predicted to have a Z disc targeting motif. [1] Filamin C is an actin binding homodimeric protein composed of two 290 kDa subunits. Each subunit is composed of an α actinin like N terminal actin binding domain (ABD) made up of 2 calponin homology tandem repeats followed by a flexible rod region containing 24 Immunoglobulin like domains (Ig- like) of around 96 residues each. The most C terminal domain (Ig 24) is the self association domain required for its dimerization ability. (Shown on right) The presence of 2 flexible calpain sensitive hinges, Hinge 1 between domain 15 and 16 divides the subunit into Rod 1 and Rod 2 domains and Hinge 2 between 23 and 24 separates the dimerization domain from the rest of domains. Each Ig domain is made of 7 β strands arranged antiparallel in group of 4 and 3 sheets forming a β sandwich. As the three Filamin proteins share around 70% homology over the entire sequence with the exception of the hinges [2], not many structures of the Filamin C domains exist in the PDB.
Sequence
Amino acid sequence of Human Filamin C is available from uniprot. However, the sequence annotation based on the tertiary structure alignment with Dictyostelium gelation factor (ABP-120) [2] is provided here.
3D StructuresSo far, only 7 3 dimensional structures of Filamin C domains exist in the protein database, out which only 2 are solved by X ray crystallography and the rest are solved by NMR. 2nqc - This is a structure of Ig-like domain 23 from human filamin C solved by X ray Crystallography 2d7q - This is a solution structure of the 23th Filamin domain from human Filamin C solved by Solution NMR 2d7m - This is a solution structure of the 14th Filamin domain from human Filamin C solved by Solution NMR 2d7n - This is a solution structure of the 16th Filamin domain from human Filamin C solved by Solution NMR 2d7o - This is a solution structure of the 17th Filamin domain from human Filamin C solved by Solution NMR 2d7p - This is a solution structure of the 22th Filamin domain from human Filamin C solved by Solution NMR 1v05 - This is a structure of the Domain 24 (Dimerization domain) of human Filamin C solved by X ray Crystallography
Functions and interaction partners of Filamin C
Since its discovery in 1975 as one of the most potent crosslinkers of F- actin [3] , major efforts have been focused to elucidate the role of Filamins as scaffolding and signaling molecule in cells. Its major functions include: • Cross linking actin filaments to form Orthogonal branched networks • Physically linking actin cytoskeleton to the membrane • Localization of the membrane receptors and stabilization of the membrane • Serving as scaffold for various interacting proteins which indicates its role in signalling
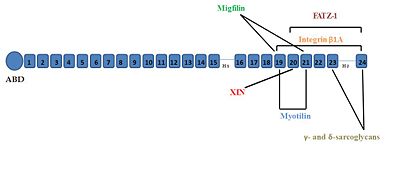
Some of the major interacting partners are shown in the diagram here. • Integrin β1A - Domain 19-24 [4] • Migfilin - Domain 21 [5] • FATZ-1 (myozenin-1, calsarcin 2) - Domain 20-24[6] • Gamma- and delta-sarcoglycans - Domain 23-24 [8]
PathologyMutations in Filamin C gene form a rare cause Myofibrillar Myopathy (MFM) presenting a wide spectrum of clinical symptoms, mostly involving progressive muscle weakness in all limbs. • First mutation identified in FLNc was in Ig domain 24 (Dimerization domain), caused by a non sense mutation of (8130G-->A; W2710X) [9] • Recently an in frame 6 amino acid deletion (Lys899-Val904) and 2 amino acid insertion (Val 899-Cys900) was identified in Ig domain 7 [10] • And another in frame 4 amino acid (Val930_Thr933) deletion mutation in Ig domain 7 has been found. [11]
References
Extra Reading
| ||||||||||||