This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Jamie Abbott/Sandbox2
From Proteopedia
(→Histidyl-tRNA Synthetase) |
(→Histidyl-tRNA Synthetase) |
||
| Line 1: | Line 1: | ||
== Histidyl-tRNA Synthetase == | == Histidyl-tRNA Synthetase == | ||
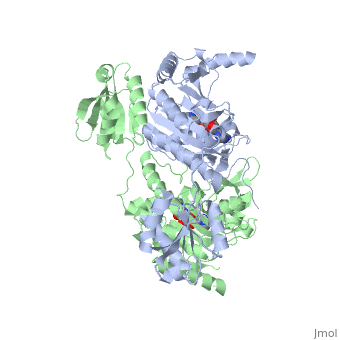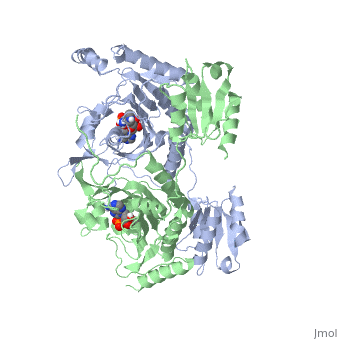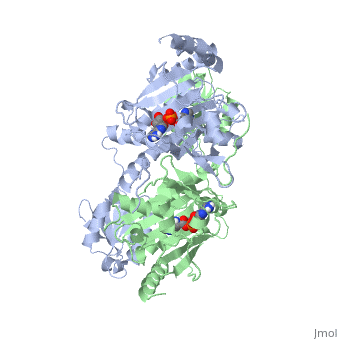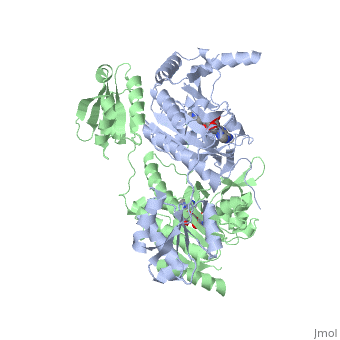
| - | Histidyl tRNA Synthetase (HisRS) is a 94kD <scene name='User:Jamie_Abbott/Sandbox2/Hisrsdimer/2'>homodimer</scene> that belongs to the Class II of aminoacyl-tRNA synthetases (aaRS). Aminoacyl-tRNA synthetases have been partitioned into two classes on the basis of sequence<ref>PMID: 2203971</ref>. Class II enzymes have a <scene name='User:Jamie_Abbott/Sandbox2/Catalytic_domain/1'>catalytic domain</scene> composed of anti-parallel β-sheets and α-helices (residues 1-325). | + | Histidyl tRNA Synthetase (HisRS) is a 94kD <scene name='User:Jamie_Abbott/Sandbox2/Hisrsdimer/2'>homodimer</scene> that belongs to the Class II of aminoacyl-tRNA synthetases (aaRS). Aminoacyl-tRNA synthetases have been partitioned into two classes, containing 10 members, on the basis of sequence comparisons<ref name="Eriani">PMID: 2203971</ref>. ClassI and Class II differ mainly with respect to the topology of the catalytic fold and site of esterification on cognate tRNA<ref name="Eriani" />. Class II enzymes have a <scene name='User:Jamie_Abbott/Sandbox2/Catalytic_domain/1'>catalytic domain</scene> composed of anti-parallel β-sheets and α-helices (residues 1-325). |
'''Function and Catalysis'''<StructureSection load='1KMM' size='500' side='right' caption='Structure of Histidyl-tRNA Synthetase (PDB entry [[1KMM]])' scene=''>Class II aminoacyl-tRNA synthetases aminoacylate the 3'OH of their cognate tRNAs. | '''Function and Catalysis'''<StructureSection load='1KMM' size='500' side='right' caption='Structure of Histidyl-tRNA Synthetase (PDB entry [[1KMM]])' scene=''>Class II aminoacyl-tRNA synthetases aminoacylate the 3'OH of their cognate tRNAs. | ||
Revision as of 21:05, 14 April 2012
Contents |
Histidyl-tRNA Synthetase
Histidyl tRNA Synthetase (HisRS) is a 94kD that belongs to the Class II of aminoacyl-tRNA synthetases (aaRS). Aminoacyl-tRNA synthetases have been partitioned into two classes, containing 10 members, on the basis of sequence comparisons[1]. ClassI and Class II differ mainly with respect to the topology of the catalytic fold and site of esterification on cognate tRNA[1]. Class II enzymes have a composed of anti-parallel β-sheets and α-helices (residues 1-325).
Function and Catalysis
| |||||||||||
Mechanism
3D Structures of Histidyl-tRNA Synthetase
References
- ↑ 1.0 1.1 Eriani G, Delarue M, Poch O, Gangloff J, Moras D. Partition of tRNA synthetases into two classes based on mutually exclusive sets of sequence motifs. Nature. 1990 Sep 13;347(6289):203-6. PMID:2203971 doi:http://dx.doi.org/10.1038/347203a0
- ↑ 2.0 2.1 Francklyn, C., and Arnez, J.G. (2004) in Aminoacyl-tRNA Synthetases (Ibba, M.,Francklyn, C.,Cusack, S.. Eds.) Landes Publishing, Austin, TX