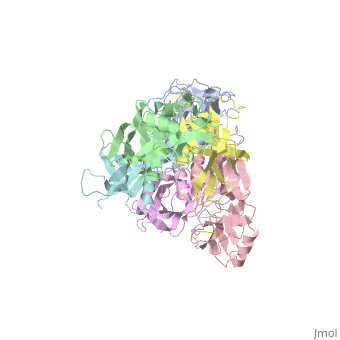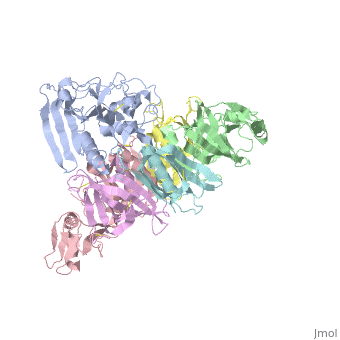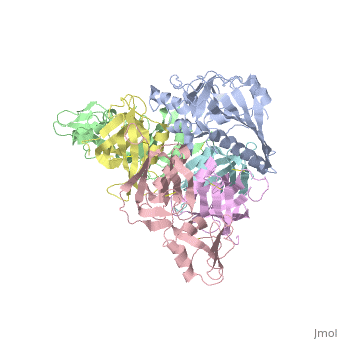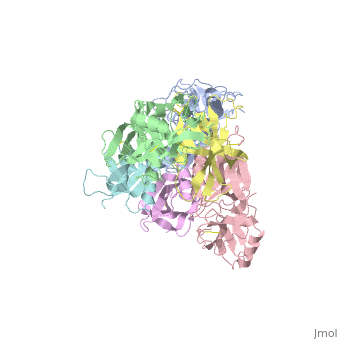This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
1prt
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
| - | [[ | + | ==THE CRYSTAL STRUCTURE OF PERTUSSIS TOXIN== |
| + | <StructureSection load='1prt' size='340' side='right' caption='[[1prt]], [[Resolution|resolution]] 2.90Å' scene=''> | ||
| + | == Structural highlights == | ||
| + | <table><tr><td colspan='2'>[[1prt]] is a 12 chain structure with sequence from [http://en.wikipedia.org/wiki/Bordetella_pertussis Bordetella pertussis]. The September 2005 RCSB PDB [http://pdb.rcsb.org/pdb/static.do?p=education_discussion/molecule_of_the_month/index.html Molecule of the Month] feature on ''Cholera Toxin'' by David S. Goodsell is [http://dx.doi.org/10.2210/rcsb_pdb/mom_2005_9 10.2210/rcsb_pdb/mom_2005_9]. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=1PRT OCA]. For a <b>guided tour on the structure components</b> use [http://oca.weizmann.ac.il/oca-docs/fgij/fg.htm?mol=1PRT FirstGlance]. <br> | ||
| + | </td></tr><tr><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[http://oca.weizmann.ac.il/oca-docs/fgij/fg.htm?mol=1prt FirstGlance], [http://oca.weizmann.ac.il/oca-bin/ocaids?id=1prt OCA], [http://www.rcsb.org/pdb/explore.do?structureId=1prt RCSB], [http://www.ebi.ac.uk/pdbsum/1prt PDBsum]</span></td></tr> | ||
| + | <table> | ||
| + | == Evolutionary Conservation == | ||
| + | [[Image:Consurf_key_small.gif|200px|right]] | ||
| + | Check<jmol> | ||
| + | <jmolCheckbox> | ||
| + | <scriptWhenChecked>select protein; define ~consurf_to_do selected; consurf_initial_scene = true; script "/wiki/ConSurf/pr/1prt_consurf.spt"</scriptWhenChecked> | ||
| + | <scriptWhenUnchecked>script /wiki/extensions/Proteopedia/spt/initialview01.spt</scriptWhenUnchecked> | ||
| + | <text>to colour the structure by Evolutionary Conservation</text> | ||
| + | </jmolCheckbox> | ||
| + | </jmol>, as determined by [http://consurfdb.tau.ac.il/ ConSurfDB]. You may read the [[Conservation%2C_Evolutionary|explanation]] of the method and the full data available from [http://bental.tau.ac.il/new_ConSurfDB/chain_selection.php?pdb_ID=2ata ConSurf]. | ||
| + | <div style="clear:both"></div> | ||
| + | <div style="background-color:#fffaf0;"> | ||
| + | == Publication Abstract from PubMed == | ||
| + | BACKGROUND: Pertussis toxin is an exotoxin of the A-B class produced by Bordetella pertussis. The holotoxin comprises 952 residues forming six subunits (five different sequences, S1-S5). It plays an important role in the development of protective immunity to whooping cough, and is an essential component of new acellular vaccines. It is also widely used as a biochemical tool to ADP-ribosylate GTP-binding proteins in the study of signal transduction. RESULTS: The crystal structure of pertussis toxin has been determined at 2.9 A resolution. The catalytic A-subunit (S1) shares structural homology with other ADP-ribosylating bacterial toxins, although differences in the carboxy-terminal portion explain its unique activation mechanism. Despite its heterogeneous subunit composition, the structure of the cell-binding B-oligomer (S2, S3, two copies of S4, and S5) resembles the symmetrical B-pentamers of the cholera toxin and Shiga toxin families, but it interacts differently with the A-subunit. The structural similarity is all the more surprising given that there is almost no sequence homology between B-subunits of the different toxins. Two peripheral domains that are unique to the pertussis toxin B-oligomer show unexpected structural homology with a calcium-dependent eukaryotic lectin, and reveal possible receptor-binding sites. CONCLUSION: The structure provides insight into the pathogenic mechanisms of pertussis toxin and the evolution of bacterial toxins. Knowledge of the tertiary structure of the active site forms a rational basis for elimination of catalytic activity in recombinant molecules for vaccine use. | ||
| - | + | The crystal structure of pertussis toxin.,Stein PE, Boodhoo A, Armstrong GD, Cockle SA, Klein MH, Read RJ Structure. 1994 Jan 15;2(1):45-57. PMID:8075982<ref>PMID:8075982</ref> | |
| - | + | From MEDLINE®/PubMed®, a database of the U.S. National Library of Medicine.<br> | |
| - | + | </div> | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
==See Also== | ==See Also== | ||
*[[Pertussis toxin|Pertussis toxin]] | *[[Pertussis toxin|Pertussis toxin]] | ||
| - | + | == References == | |
| - | == | + | <references/> |
| - | < | + | __TOC__ |
| + | </StructureSection> | ||
[[Category: Bordetella pertussis]] | [[Category: Bordetella pertussis]] | ||
[[Category: Cholera Toxin]] | [[Category: Cholera Toxin]] | ||
Revision as of 23:50, 28 September 2014
THE CRYSTAL STRUCTURE OF PERTUSSIS TOXIN
| |||||||||||