This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Josie N. Harmon/Sandbox Tutorial
From Proteopedia
(Difference between revisions)
| Line 11: | Line 11: | ||
== Mechanism of Action == | == Mechanism of Action == | ||
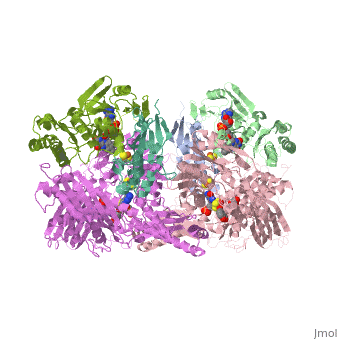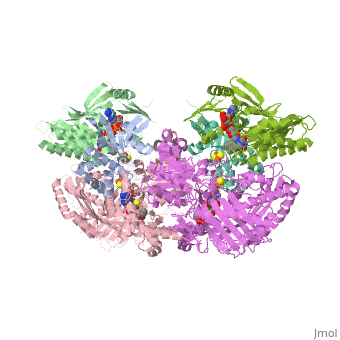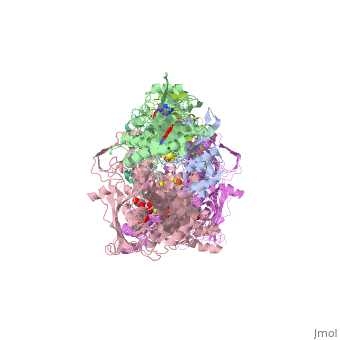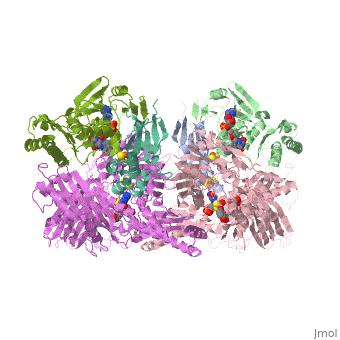
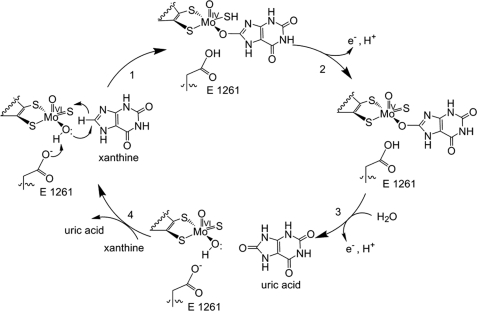
| - | Xanthine oxidase is characterized as a molybdenum containing enzyme that catalyzes the hydroxylation of a sp2 hybrized carbon in a broad range of aromatic heterocycles and aldehydes. In eukaryotes xanthine oxidase exist as a homodimer with each monomer containing four redox-active sites. The crystal structure of the bovine xanthine oxidase complex contains two active sites with varying intrinsic activity. The crystalline structure of a xanthine oxidase monomer offers a better view of the active molybdenum center, the ferredoxin iron sulfur, Fe2S2, clusters, and FAD. The <scene name='User:Josie_N._Harmon/Sandbox_1/Active_site/1'>active site</scene> is thought to be composed of glutamine, glutamic acid, phenylalanine, arginine, and molybdenum center. The substrate is believed to bind between the Phe 1009 and Phe 914. | + | Xanthine oxidase is characterized as a molybdenum containing enzyme that catalyzes the hydroxylation of a sp2 hybrized carbon in a broad range of aromatic heterocycles and aldehydes. In eukaryotes xanthine oxidase exist as a homodimer with each monomer containing four redox-active sites. The crystal structure of the bovine xanthine oxidase complex contains two active sites with varying intrinsic activity. The crystalline structure of a xanthine oxidase monomer offers a better view of the active molybdenum center, the ferredoxin iron sulfur, Fe2S2, clusters, and FAD. The <scene name='User:Josie_N._Harmon/Sandbox_1/Active_site/1'>active site</scene> is thought to be composed of glutamine, glutamic acid, phenylalanine, arginine, and the molybdenum center. The substrate is believed to bind between the Phe 1009 and Phe 914. |
== Electron Extraction == | == Electron Extraction == | ||
Revision as of 19:16, 12 November 2012
Xanthine Oxidase Biochemistry Tutorial
The purpose of this tutorial is to explain the mechanism of the metabolic enzyme xanthine oxidoreductase.
| |||||||||||