This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 390
From Proteopedia
(Difference between revisions)
| Line 14: | Line 14: | ||
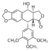
In this paper, the researchers reported on the crystal structure of a large fragment of type II human topoisomerases β (hTOP2β core) complexed to DNA and to the anticancer drug etoposide to reveal structural details of drug-induced stabilization of a cleavage complex<ref>PMID: 21778401</ref>. This structure provided the first observation of a TOP2 ternary cleavage complex <scene name='Sandbox_Reserved_390/Top/22'>stabilized</scene> by an anticancer drug. | In this paper, the researchers reported on the crystal structure of a large fragment of type II human topoisomerases β (hTOP2β core) complexed to DNA and to the anticancer drug etoposide to reveal structural details of drug-induced stabilization of a cleavage complex<ref>PMID: 21778401</ref>. This structure provided the first observation of a TOP2 ternary cleavage complex <scene name='Sandbox_Reserved_390/Top/22'>stabilized</scene> by an anticancer drug. | ||
| - | The high-resolution structure of the hTOP2βcore-DNA-etoposide ternary complex reveals the intricate interplays between <scene name='Sandbox_Reserved_390/Top/6'>protein</scene>, <scene name='Sandbox_Reserved_390/Top/16'>DNA</scene> and <scene name='Sandbox_Reserved_390/Top/ | + | The high-resolution structure of the hTOP2βcore-DNA-etoposide ternary complex reveals the intricate interplays between <scene name='Sandbox_Reserved_390/Top/6'>protein</scene>, <scene name='Sandbox_Reserved_390/Top/16'>DNA</scene> and <scene name='Sandbox_Reserved_390/Top/23'>drugs</scene>. This aspect is extremely important because all vertebrates possess two highly similar yet functionally distinct TOP2 isoforms. The α-isoform is particularly important for DNA replication and is usually present at high levels in fast growing cancer cells, whereas the β-isoform is mainly involved in transcription related processes. Although the inhibition of both TOP2 isoforms contributes to the drug-induced death of cancer cells, targeting of the β-isoform has been implicated in deleterious therapy related secondary malignancies. Therefore, it is desirable to develop the isoform specific TOP2-targeting agents. |
==COMPOUND ACTIVE SITE== | ==COMPOUND ACTIVE SITE== | ||
Revision as of 22:28, 19 November 2012
Human topoisomerase IIbeta in complex with DNA and etoposide
| |||||||||||
References
- ↑ Wu CC, Li TK, Farh L, Lin LY, Lin TS, Yu YJ, Yen TJ, Chiang CW, Chan NL. Structural basis of type II topoisomerase inhibition by the anticancer drug etoposide. Science. 2011 Jul 22;333(6041):459-62. PMID:21778401 doi:10.1126/science.1204117
- ↑ Kathryn L. Gilroy, Chrysoula Leontiou, Kay Padget, Jeremy H. Lakey and Caroline A. Austin* "mAMSA resistant human topoisomerase IIβ mutation G465D has reduced ATP hydrolysis activity” Oxford JournalsLife Sciences Nucleic Acids Research Volume 34, Issue 5Pp. 1597-1607. DOI: 10.1093/nar/gkl057
- ↑ Wu CC, Li TK, Farh L, Lin LY, Lin TS, Yu YJ, Yen TJ, Chiang CW, Chan NL. Structural basis of type II topoisomerase inhibition by the anticancer drug etoposide. Science. 2011 Jul 22;333(6041):459-62. PMID:21778401 doi:10.1126/science.1204117