Sandbox Reserved 390
From Proteopedia
(Difference between revisions)
| Line 10: | Line 10: | ||
Type II topoisomerases (TOP2s) are abundant enzymes that play an essential role in <scene name='Sandbox_Reserved_390/Top/5'>DNA</scene> replication and transcription and are important targets for cancer chemotherapeutic drugs. These enzymes briefly cleave a pair of opposing phosphodiester bonds four base pairs apart, generating a TOP2-DNA cleavage complex. | Type II topoisomerases (TOP2s) are abundant enzymes that play an essential role in <scene name='Sandbox_Reserved_390/Top/5'>DNA</scene> replication and transcription and are important targets for cancer chemotherapeutic drugs. These enzymes briefly cleave a pair of opposing phosphodiester bonds four base pairs apart, generating a TOP2-DNA cleavage complex. | ||
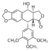
| - | TOP2’s DNA cleavage activity is usually referred to as a double-edged sword; failure to reseal the enzyme-mediated DNA break can lead to cell death. Several potent anticancer drugs, such as<scene name='Sandbox_Reserved_390/ | + | TOP2’s DNA cleavage activity is usually referred to as a double-edged sword; failure to reseal the enzyme-mediated DNA break can lead to cell death. Several potent anticancer drugs, such as <scene name='Sandbox_Reserved_390/Etoposide/1'>etoposide</scene> , <scene name='Sandbox_Reserved_390/Doxorubicin/1'>doxorubicin</scene> and <scene name='Sandbox_Reserved_390/Mitoxantron/1'>mitoxantrone</scene> (in green), exploit this harmful aspect of TOP2 and promote the formation of cytotoxic DNA lesions by increasing the stability level of cleavage complexes. <ref> Kathryn L. Gilroy, Chrysoula Leontiou, Kay Padget, Jeremy H. Lakey and Caroline A. Austin* "mAMSA resistant human topoisomerase IIβ mutation G465D has reduced ATP hydrolysis activity” Oxford JournalsLife Sciences Nucleic Acids Research Volume 34, Issue 5Pp. 1597-1607. [http://nar.oxfordjournals.org/content/34/5/1597 DOI: 10.1093/nar/gkl057]</ref> |
In this paper, the researchers reported on the crystal structure of a large fragment of type II human topoisomerases β (hTOP2β core) complexed to DNA and to the anticancer drug etoposide to reveal structural details of drug-induced stabilization of a cleavage complex<ref>PMID: 21778401</ref>. This structure provided the first observation of a TOP2 ternary cleavage complex <scene name='Sandbox_Reserved_390/Top/22'>stabilized</scene> by an anticancer drug. | In this paper, the researchers reported on the crystal structure of a large fragment of type II human topoisomerases β (hTOP2β core) complexed to DNA and to the anticancer drug etoposide to reveal structural details of drug-induced stabilization of a cleavage complex<ref>PMID: 21778401</ref>. This structure provided the first observation of a TOP2 ternary cleavage complex <scene name='Sandbox_Reserved_390/Top/22'>stabilized</scene> by an anticancer drug. | ||
Revision as of 17:56, 20 November 2012
Human topoisomerase IIbeta in complex with DNA and etoposide
| |||||||||||
References
- ↑ Wu CC, Li TK, Farh L, Lin LY, Lin TS, Yu YJ, Yen TJ, Chiang CW, Chan NL. Structural basis of type II topoisomerase inhibition by the anticancer drug etoposide. Science. 2011 Jul 22;333(6041):459-62. PMID:21778401 doi:10.1126/science.1204117
- ↑ Kathryn L. Gilroy, Chrysoula Leontiou, Kay Padget, Jeremy H. Lakey and Caroline A. Austin* "mAMSA resistant human topoisomerase IIβ mutation G465D has reduced ATP hydrolysis activity” Oxford JournalsLife Sciences Nucleic Acids Research Volume 34, Issue 5Pp. 1597-1607. DOI: 10.1093/nar/gkl057
- ↑ Wu CC, Li TK, Farh L, Lin LY, Lin TS, Yu YJ, Yen TJ, Chiang CW, Chan NL. Structural basis of type II topoisomerase inhibition by the anticancer drug etoposide. Science. 2011 Jul 22;333(6041):459-62. PMID:21778401 doi:10.1126/science.1204117