This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Fragment-Based Drug Discovery
From Proteopedia
(Difference between revisions)
| Line 13: | Line 13: | ||
| - | The development of <scene name='Sandbox_reserved_394/Abt-737/ | + | The development of <scene name='Sandbox_reserved_394/Abt-737/5'>ABT-737</scene> using SAR by NMR is a classic example of FBDD. (Throughout this discussion ABT-737 will be used to illustrate the FBDD process.) This compound has been shown to effectively inhibit the over-expression of <scene name='Sandbox_reserved_394/Bcl-xl/1'>Bcl-xl</scene> which is a protein that is commonly observed to be over-expressed in many types of cancers.<ref name="Oltersdorf T., Elmore S. W., Shoemaker A. R. An inhibitor of Bcl-2 family proteins induces regression of solid tumours. Vol 435|2 June 2005|doi:10.1038/nature03579">Oltersdorf T., Elmore S. W., Shoemaker A. R. An inhibitor of Bcl-2 family proteins induces regression of solid tumours. Vol 435|2 June 2005|doi:10.1038/nature03579</ref> It acts an inhibitor of apoptosis and may also contribute to chemotherapy resistance. Bcl-xl inhibition by ABT-737 therefore, allows apoptosis to occur and helps to prevent chemo-resistance. |
{| class="wikitable collapsible" | {| class="wikitable collapsible" | ||
! scope="col" width="5000px" | SAR by NMR | ! scope="col" width="5000px" | SAR by NMR | ||
| Line 26: | Line 26: | ||
===== ABT-737: ligand screening ===== | ===== ABT-737: ligand screening ===== | ||
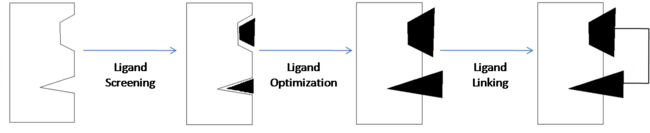
| - | <scene name='Sandbox_reserved_394/Compound_1/ | + | <scene name='Sandbox_reserved_394/Compound_1/12'>Two fragments</scene> were found to have moderate affinity for Bcl-xl. <scene name='Sandbox_reserved_394/Compound_1/9'>Compound 1</scene> is a fluorobiphenylcarboxylic acid. It occupies <scene name='Sandbox_reserved_394/Binding_site_1/2'>binding site 1</scene> of Bcl-xl which consists of Phe 101, Tyr 105, Ala 108, Phe 109, Leu 136, Gly 142, Arg 143, and Ala 146. The fluorobiphenyl system of compound 1 is very hydrophobic and therefore, these residues form a <scene name='Sandbox_reserved_394/Compound_1/4'>"hydrophobic pocket"</scene> around the system. There is also one hydrophilic interaction involved in this complex. The <scene name='Sandbox_reserved_394/Compound_1/5'>carboxylic acid portion of compound 1 binds near Gly 142</scene> of Bcl-xl. This is not a strong interaction but is significant because it can be modified to form a much stronger bond. |
<scene name='Sandbox_reserved_394/Compound_1/3'>Compound 2</scene> is a napthalene-based alcohol which occupies <scene name='Sandbox_reserved_394/Binding_site_2/4'>binding site 2</scene>. This particular fragment also is involved with hydrophobic interactions with Bcl-xl, although they are not as strong as in the case of compound 1. This binding site includes Ala 97, Glu 100, Phe 101, Val 145, and Tyr 199. | <scene name='Sandbox_reserved_394/Compound_1/3'>Compound 2</scene> is a napthalene-based alcohol which occupies <scene name='Sandbox_reserved_394/Binding_site_2/4'>binding site 2</scene>. This particular fragment also is involved with hydrophobic interactions with Bcl-xl, although they are not as strong as in the case of compound 1. This binding site includes Ala 97, Glu 100, Phe 101, Val 145, and Tyr 199. | ||
Revision as of 20:29, 1 December 2012
Drug Design: Fragment-Based Drug Discovery
| |||||||||||
References
- ↑ 1.0 1.1 Shuker S. B., Hajduk P. J., Meadows R. P., Fesik S. W. Discovering High-Affinity Ligands for Proteins: SAR by NMR. Science; Nov 29, 1996; 274, 5292; ProQuest Central pg. 1531.
- ↑ Oltersdorf T., Elmore S. W., Shoemaker A. R. An inhibitor of Bcl-2 family proteins induces regression of solid tumours. Vol 435|2 June 2005|doi:10.1038/nature03579
- ↑ Pandit D. LIGAND-BASED DRUG DESIGN: I. CONFORMATIONAL STUDIES OF GBR 12909 ANALOGS AS COCAINE ANTAGONISTS; II. 3D-QSAR STUDIES OF SALVINORIN A ANALOGS AS εΑΡΡΑ OPIOID AGONISTS. http://archives.njit.edu/vol01/etd/2000s/2007/njit-etd2007-051/njit-etd2007-051.pdf