This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox UC 9
From Proteopedia
(Difference between revisions)
| Line 4: | Line 4: | ||
<Structure load='1pgb' size='300' frame='true' align='right' caption=' Torpedo acetylcholinesterase' scene='Insert optional scene name here' /> | <Structure load='1pgb' size='300' frame='true' align='right' caption=' Torpedo acetylcholinesterase' scene='Insert optional scene name here' /> | ||
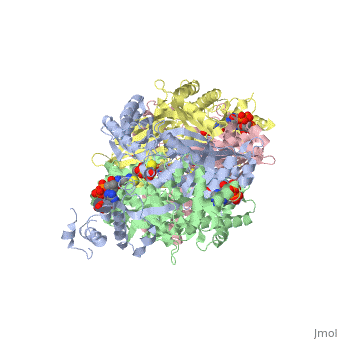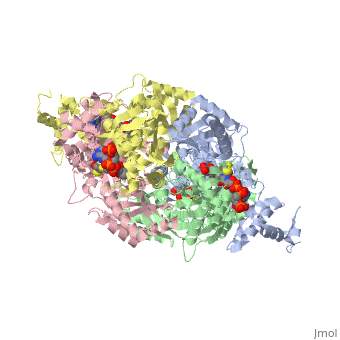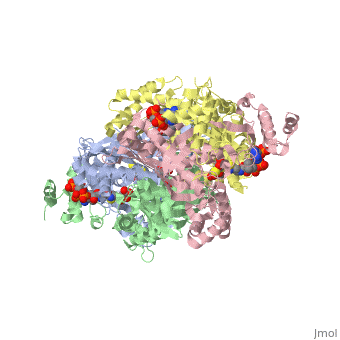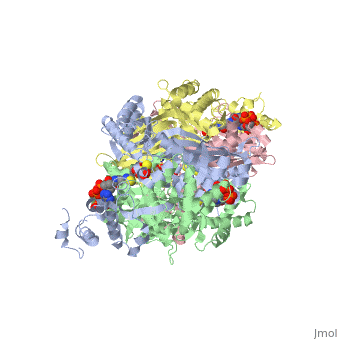
| - | PubMed Abstract: | + | ==Your Heading Here (maybe something like 'Structure')==<StructureSection load='1dq8' size='500' side='right' caption='Structure of HMG-CoA reductase (PDB entry [[1dq8]])' scene=''>PubMed Abstract: |
| - | Binding sites of Torpedo acetylcholinesterase (EC 3.1.1.7) for quaternary ligands were investigated by x-ray crystallography and photoaffinity labeling. Crystal structures of complexes with ligands were determined at 2.8-A resolution. In a complex with edrophonium, and quaternary nitrogen of the ligand interacts with the indole of Trp-84, and its m-hydroxyl displays bifurcated hydrogen bonding to two members of the catalytic triad, Ser-200 and His-440. In a complex with tacrine, the acridine is stacked against the indole of Trp-84. The bisquaternary ligand decamethonium is oriented along the narrow gorge leading to the active site; one quaternary group is apposed to the indole of Trp-84 and the other to that of Trp-279, near the top of the gorge. The only major conformational difference between the three complexes is in the orientation of the phenyl ring of Phe-330. In the decamethonium complex it lies parallel to the surface of the gorge; in the other two complexes it is positioned to make contact with the bound ligand. This close interaction was confirmed by photoaffinity labelling by the photosensitive probe 3H-labeled p-(N,N-dimethylamino)benzenediazonium fluoroborate, which labeled, predominantly, Phe-330 within the active site. Labeling of Trp-279 was also observed. One mole of label is incorporated per mole of AcChoEase inactivated, indicating that labeling of Trp-279 and that of Phe-330 are mutually exclusive. The structural and chemical data, together, show the important role of aromatic groups as binding sites for quaternary ligands, and they provide complementary evidence assigning Trp-84 and Phe-330 to the "anionic" subsite of the active site and Trp-279 to the "peripheral" anionic site. | + | Binding sites of Torpedo acetylcholinesterase (EC 3.1.1.7) for quaternary ligands were investigated by x-ray crystallography and photoaffinity labeling. Crystal structures of complexes with ligands were determined at 2.8-A resolution. In a complex with edrophonium, and quaternary nitrogen of the ligand interacts with the indole of Trp-84, and its m-hydroxyl displays bifurcated hydrogen bonding to two members of the catalytic triad, Ser-200 and His-440. In a complex with tacrine, the acridine is stacked against the indole of Trp-84. The bisquaternary ligand decamethonium is oriented along the narrow gorge leading to the active site; one quaternary group is apposed to the indole of Trp-84 and the other to that of Trp-279, near the top of the gorge. The only major conformational difference between the three complexes is in the orientation of the phenyl ring of Phe-330. In the decamethonium complex it lies parallel to the surface of the gorge; in the other two complexes it is positioned to make contact with the bound ligand. This close interaction was confirmed by photoaffinity labelling by the photosensitive probe 3H-labeled p-(N,N-dimethylamino)benzenediazonium fluoroborate, which labeled, predominantly, Phe-330 within the active site. Labeling of Trp-279 was also observed. One mole of label is incorporated per mole of AcChoEase inactivated, indicating that labeling of Trp-279 and that of Phe-330 are mutually exclusive. The structural and chemical data, together, show the important role of aromatic groups as binding sites for quaternary ligands, and they provide complementary evidence assigning Trp-84 and Phe-330 to the "anionic" subsite of the active site and Trp-279 to the "peripheral" anionic site.</StructureSection> |
[[Image:MW_Folding_Simulations.gif]] | [[Image:MW_Folding_Simulations.gif]] | ||
| Line 16: | Line 16: | ||
{{Template:ColorKey_Amino2CarboxyRainbow}} | {{Template:ColorKey_Amino2CarboxyRainbow}} | ||
| - | <quiz display=simple> | ||
| - | {que dia es hoy?... | ||
| - | |type="()"} | ||
| - | - lunes | ||
| - | + miercoles | ||
| - | - sabado | ||
| - | </quiz> | ||
Revision as of 20:35, 19 December 2012
Quaternary ligand binding to aromatic residues in the active-site gorge of acetylcholinesterase.
|
| |||||||||||
lalalala
| Amino Terminus | Carboxy Terminus |
|
| |||||||||
| 1acj, resolution 2.80Å () | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Ligands: | |||||||||
| Activity: | Acetylcholinesterase, with EC number 3.1.1.7 | ||||||||
| |||||||||
| |||||||||
| Resources: | FirstGlance, OCA, RCSB, PDBsum | ||||||||
| Coordinates: | save as pdb, mmCIF, xml | ||||||||