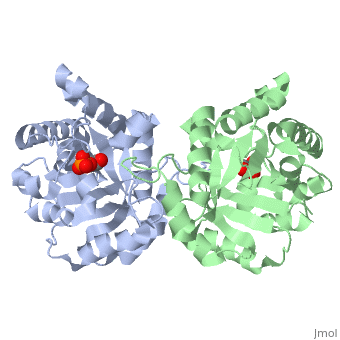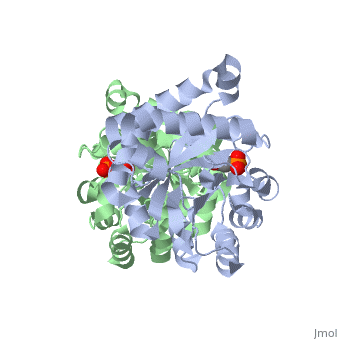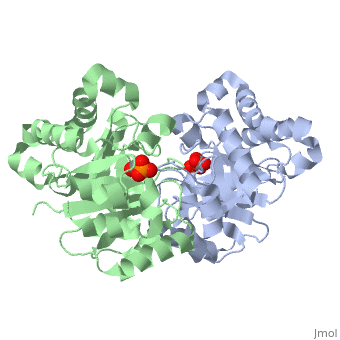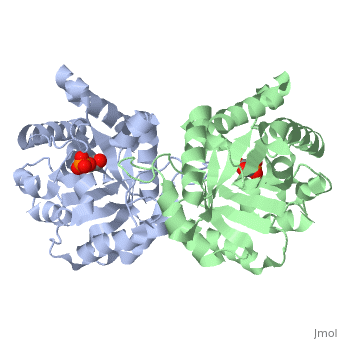This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
2ypi
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
| - | + | ==CRYSTALLOGRAPHIC ANALYSIS OF THE COMPLEX BETWEEN TRIOSEPHOSPHATE ISOMERASE AND 2-PHOSPHOGLYCOLATE AT 2.5-ANGSTROMS RESOLUTION. IMPLICATIONS FOR CATALYSIS== | |
| - | + | <StructureSection load='2ypi' size='340' side='right' caption='[[2ypi]], [[Resolution|resolution]] 2.50Å' scene=''> | |
| - | + | == Structural highlights == | |
| + | <table><tr><td colspan='2'>[[2ypi]] is a 2 chain structure with sequence from [http://en.wikipedia.org/wiki/Saccharomyces_cerevisiae Saccharomyces cerevisiae]. The February 2004 RCSB PDB [http://pdb.rcsb.org/pdb/static.do?p=education_discussion/molecule_of_the_month/index.html Molecule of the Month] feature on ''The Glycolytic Enzymes'' by David S. Goodsell is [http://dx.doi.org/10.2210/rcsb_pdb/mom_2004_2 10.2210/rcsb_pdb/mom_2004_2]. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=2YPI OCA]. For a <b>guided tour on the structure components</b> use [http://oca.weizmann.ac.il/oca-docs/fgij/fg.htm?mol=2YPI FirstGlance]. <br> | ||
| + | </td></tr><tr><td class="sblockLbl"><b>[[Ligand|Ligands:]]</b></td><td class="sblockDat"><scene name='pdbligand=PGA:2-PHOSPHOGLYCOLIC+ACID'>PGA</scene><br> | ||
| + | <tr><td class="sblockLbl"><b>Activity:</b></td><td class="sblockDat"><span class='plainlinks'>[http://en.wikipedia.org/wiki/Triose-phosphate_isomerase Triose-phosphate isomerase], with EC number [http://www.brenda-enzymes.info/php/result_flat.php4?ecno=5.3.1.1 5.3.1.1] </span></td></tr> | ||
| + | <tr><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[http://oca.weizmann.ac.il/oca-docs/fgij/fg.htm?mol=2ypi FirstGlance], [http://oca.weizmann.ac.il/oca-bin/ocaids?id=2ypi OCA], [http://www.rcsb.org/pdb/explore.do?structureId=2ypi RCSB], [http://www.ebi.ac.uk/pdbsum/2ypi PDBsum]</span></td></tr> | ||
| + | <table> | ||
| + | == Evolutionary Conservation == | ||
| + | [[Image:Consurf_key_small.gif|200px|right]] | ||
| + | Check<jmol> | ||
| + | <jmolCheckbox> | ||
| + | <scriptWhenChecked>select protein; define ~consurf_to_do selected; consurf_initial_scene = true; script "/wiki/ConSurf/yp/2ypi_consurf.spt"</scriptWhenChecked> | ||
| + | <scriptWhenUnchecked>script /wiki/extensions/Proteopedia/spt/initialview01.spt</scriptWhenUnchecked> | ||
| + | <text>to colour the structure by Evolutionary Conservation</text> | ||
| + | </jmolCheckbox> | ||
| + | </jmol>, as determined by [http://consurfdb.tau.ac.il/ ConSurfDB]. You may read the [[Conservation%2C_Evolutionary|explanation]] of the method and the full data available from [http://bental.tau.ac.il/new_ConSurfDB/chain_selection.php?pdb_ID=2ata ConSurf]. | ||
| + | <div style="clear:both"></div> | ||
| + | <div style="background-color:#fffaf0;"> | ||
| + | == Publication Abstract from PubMed == | ||
| + | The binding of the transition-state analogue 2-phosphoglycolate to triosephosphate isomerase from yeast has been investigated crystallographically. An atomic model of the enzyme-inhibitor complex has been refined against data to 2.5-A resolution to a final R factor of 0.18. The interactions between the inhibitor and enzyme have been analyzed. The inhibitor forms hydrogen bonds to the side chains of His 95 and Glu 165. The latter hydrogen bond confirms that Glu 165 is protonated upon PGA binding. The structure of the complexed enzyme has been compared to that of the unbound form of the enzyme, and conformational changes have been observed: the side chain of Glu 165 moves over 2 A and a 10-residue flexible loop moves over 7 A to close over the active site. Spectroscopic results of phosphoglycolic acid binding to triosephosphate isomerase that have been amassed over the years are also explained in structural terms. The implications for catalysis are noted. | ||
| - | + | Crystallographic analysis of the complex between triosephosphate isomerase and 2-phosphoglycolate at 2.5-A resolution: implications for catalysis.,Lolis E, Petsko GA Biochemistry. 1990 Jul 17;29(28):6619-25. PMID:2204418<ref>PMID:2204418</ref> | |
| - | + | ||
| + | From MEDLINE®/PubMed®, a database of the U.S. National Library of Medicine.<br> | ||
| + | </div> | ||
==See Also== | ==See Also== | ||
*[[Triose Phosphate Isomerase|Triose Phosphate Isomerase]] | *[[Triose Phosphate Isomerase|Triose Phosphate Isomerase]] | ||
| - | + | == References == | |
| - | == | + | <references/> |
| - | < | + | __TOC__ |
| + | </StructureSection> | ||
[[Category: RCSB PDB Molecule of the Month]] | [[Category: RCSB PDB Molecule of the Month]] | ||
[[Category: Saccharomyces cerevisiae]] | [[Category: Saccharomyces cerevisiae]] | ||
Revision as of 05:11, 2 October 2014
CRYSTALLOGRAPHIC ANALYSIS OF THE COMPLEX BETWEEN TRIOSEPHOSPHATE ISOMERASE AND 2-PHOSPHOGLYCOLATE AT 2.5-ANGSTROMS RESOLUTION. IMPLICATIONS FOR CATALYSIS
| |||||||||||