We apologize for Proteopedia being slow to respond. For the past two years, a new implementation of Proteopedia has been being built. Soon, it will replace this 18-year old system. All existing content will be moved to the new system at a date that will be announced here.
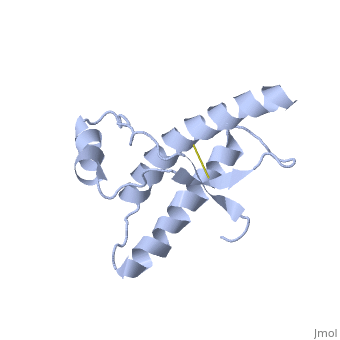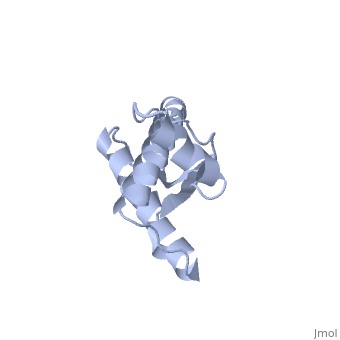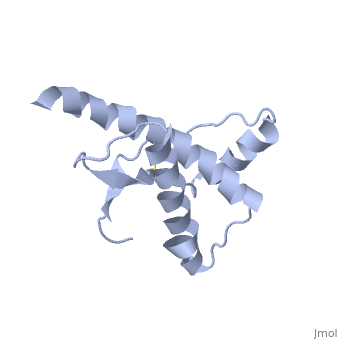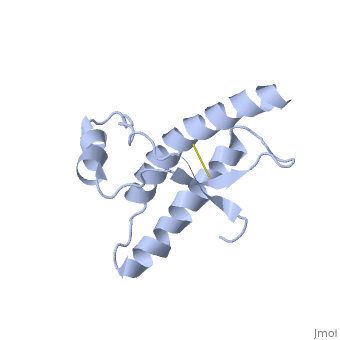
Human Prion Protein Dimer
From Proteopedia
(Difference between revisions)
| Line 29: | Line 29: | ||
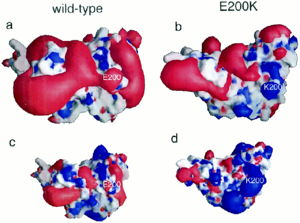
| - | [[Image:F4.large.jpg| | + | [[Image:F4.large.jpg|left|300px|thumb|Electrostatic potential alteration E200K|Caption: This shows (a&c) the electrostatic potential of wild-type Human Prion Protein with Glu200 and (b&d) the electrostatic potential of variant Lys200. <ref name="Zhang">PMID:10954699</ref>]] |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
=== PrP<sup>Sc</sup> === | === PrP<sup>Sc</sup> === | ||
| Line 54: | Line 43: | ||
The Cystine residues which were formerly part of disulfide bonds have been reduced catalytically without any chemical reducing agent. <ref name="Knaus">PMID:11524679</ref> | The Cystine residues which were formerly part of disulfide bonds have been reduced catalytically without any chemical reducing agent. <ref name="Knaus">PMID:11524679</ref> | ||
| - | |||
| - | |||
| - | </StructureSection> | ||
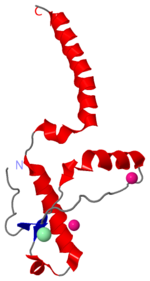
===Dimer Form=== | ===Dimer Form=== | ||
Revision as of 09:58, 8 May 2013
| |||||||||||
Reference List
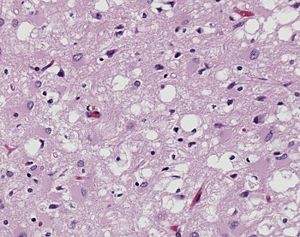
- ↑ Image of Creutzfeldt-Jakob positive brain tissue was obtained from The CDC's Public Health Image Library.
- ↑ Centers for Disease Control and Prevention: Prions. http://www.cdc.gov/ncidod/dvrd/prions/
- ↑ 3.0 3.1 Prusiner SB. Prions. Proc Natl Acad Sci U S A. 1998 Nov 10;95(23):13363-83. PMID:9811807
- ↑ Centers for Disease Control and Prevention: Bovine Spongiform Encephalopathy. http://www.cdc.gov/ncidod/dvrd/bse/index.htm
- ↑ 5.0 5.1 5.2 5.3 Lee S, Antony L, Hartmann R, Knaus KJ, Surewicz K, Surewicz WK, Yee VC. Conformational diversity in prion protein variants influences intermolecular beta-sheet formation. EMBO J. 2010 Jan 6;29(1):251-62. Epub 2009 Nov 19. PMID:19927125 doi:10.1038/emboj.2009.333
- ↑ 6.0 6.1 6.2 6.3 6.4 Knaus KJ, Morillas M, Swietnicki W, Malone M, Surewicz WK, Yee VC. Crystal structure of the human prion protein reveals a mechanism for oligomerization. Nat Struct Biol. 2001 Sep;8(9):770-4. PMID:11524679 doi:10.1038/nsb0901-770
- ↑ 7.0 7.1 7.2 7.3 Zhang Y, Swietnicki W, Zagorski MG, Surewicz WK, Sonnichsen FD. Solution structure of the E200K variant of human prion protein. Implications for the mechanism of pathogenesis in familial prion diseases. J Biol Chem. 2000 Oct 27;275(43):33650-4. PMID:10954699 doi:10.1074/jbc.C000483200
Proteopedia Page Contributors and Editors (what is this?)
Erin May, Alexander Berchansky, Jaime Prilusky, Michal Harel