This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Succinyl-CoA synthetase
From Proteopedia
| Line 1: | Line 1: | ||
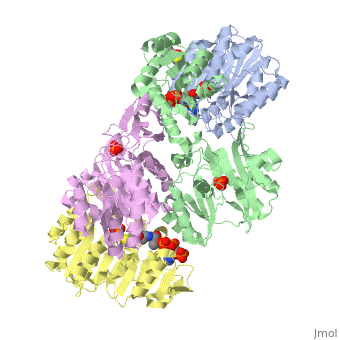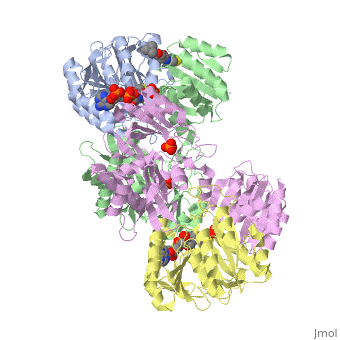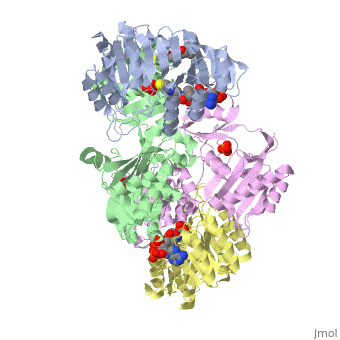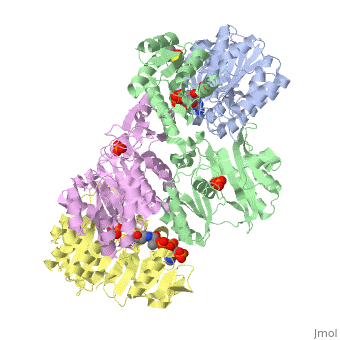
| - | <StructureSection load='1cqj' size='450' side='right' scene='' caption=''> | + | <StructureSection load='1cqj' size='450' side='right' scene='' caption='E. coli succinyl-CoA synthetase α subunit (grey, yellow) and β subunit (green, magenta) complex with CoA and phosphate (PDB code [[1cqj]])'> |
'''Succinyl-Coa synthetase''' catalyzes the reversible reaction of succinyl-CoA + NDP + Pi <-> succinate + CoA + NTP (where N is either adenosine or guanosine. It can be found in Escherichia coli and is the fifth step in the [[Citric Acid Cycle|citric acid cycle]]. Due to its involvement in the citric acid cycle it is very important in all living cells that use oxygen as a part of cellular respiration. This entire process (Citric acid cycle) is necessary in producing one GTP or ATP, three NADHs, and two carbon dioxides. | '''Succinyl-Coa synthetase''' catalyzes the reversible reaction of succinyl-CoA + NDP + Pi <-> succinate + CoA + NTP (where N is either adenosine or guanosine. It can be found in Escherichia coli and is the fifth step in the [[Citric Acid Cycle|citric acid cycle]]. Due to its involvement in the citric acid cycle it is very important in all living cells that use oxygen as a part of cellular respiration. This entire process (Citric acid cycle) is necessary in producing one GTP or ATP, three NADHs, and two carbon dioxides. | ||
Revision as of 08:20, 13 August 2013
| |||||||||||
3D structures of succinyl-CoA synthetase
Updated February 2013
Histidine-phosphorylated succinyl-CoA synthetase
1scu, 2scu – EcSCS α+β subunit – Escherichia coli
2nu6, 2nu7, 2nu8, 2nu9, 2nua - EcSCS α (mutant) + β subunit
1eud – pSCS α+β (mutant) subunit - pig
2fp4 - pSCS α+β subunit + Mg + GTP
2fpi, 2fpp - pSCS α+β subunit
Dephosphorylated succinyl-CoA synthetase
1cqi - EcSCS α+β subunit + Mg + ADP + PO4
1cqj, 1jkj - EcSCS α+β subunit + PO4
1jll - EcSCS α+β (mutant) subunit + PO4
1euc – pSCS α+β (mutant) subunit + PO4
2fpg – pSCS α+β subunit + PO4 + GDP
1oi7 – SCS α subunit (mutant) – Thermos thermophilus
2yv1 - SCS α subunit – Methanocaldococcus jannaschii
2yv2 - SCS α subunit – Aeropyrum pernix
3ufx - SCS α+β subunit + Mn + GDP – Thermus aquaticus
Additional Resources
For Additional Information, See Carbohydrate Metabolism
References
- ↑ Joyce MA, Fraser ME, Brownie ER, James MN, Bridger WA, Wolodko WT. Probing the nucleotide-binding site of Escherichia coli succinyl-CoA synthetase. Biochemistry. 1999 Jun 1;38(22):7273-83. PMID:10353839 doi:10.1021/bi990527s
- ↑ Fraser ME, Joyce MA, Ryan DG, Wolodko WT. Two glutamate residues, Glu 208 alpha and Glu 197 beta, are crucial for phosphorylation and dephosphorylation of the active-site histidine residue in succinyl-CoA synthetase. Biochemistry. 2002 Jan 15;41(2):537-46. PMID:11781092
- ↑ Bild GS, Janson CA, Boyer PD. Subunit interaction during catalysis. ATP modulation of catalytic steps in the succinyl-CoA synthetase reaction. J Biol Chem. 1980 Sep 10;255(17):8109-15. PMID:6997289
- ↑ Mikeladze DG, Matveeva LN, Severin SE. [Reaction mechanism of succinyl CoA synthetase from pigeon thoracic muscle] Biokhimiia. 1978 Aug;43(8):1458-67. PMID:570066
- ↑ Joyce MA, Fraser ME, James MN, Bridger WA, Wolodko WT. ADP-binding site of Escherichia coli succinyl-CoA synthetase revealed by x-ray crystallography. Biochemistry. 2000 Jan 11;39(1):17-25. PMID:10625475
- ↑ Frank J. Moffet and W. A. Bridger. The Kinetics of Succinyl Coenzyme A Synthetase from Escherichia coli : A REACTION WITH A COVALENT ENZYME-SUBSTRATE INTERMEDIATE NOT EXHIBITING "PING-PONG" KINETICS J. Biol. Chem. 1970 245: 2758-2762.[1]
- ↑ Um, H.-D., and C. Klein. 1993. Evidence for allosteric regulation of succinyl-CoA synthetase. Biochem. J. 295:821–826.[2]
Content Donators
Proteopedia Page Contributors and Editors (what is this?)
Michal Harel, Lucas Hamlow, J.D. McClintic, Wayne Decatur, David Canner, Alexander Berchansky