This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox reserved 916
From Proteopedia
(Difference between revisions)
| Line 3: | Line 3: | ||
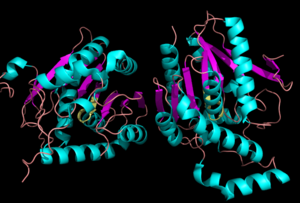
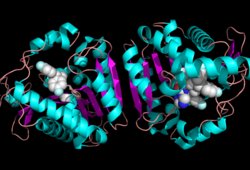
[[Image:Complete_crystal_structure.png|left|300px|thumb|Crystal Structure of MGL]] | [[Image:Complete_crystal_structure.png|left|300px|thumb|Crystal Structure of MGL]] | ||
==Background== | ==Background== | ||
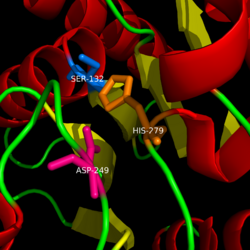
| + | Monoglyceride lipase is part of the α/β hydrolase family, having a Ser-His-Asp catalytic triad (Celemnte et al. 2012). MGL terminates the signaling of a primary endocannabinoid, 2-AG (Savinainen et al 2010). MGL is able to hydrolyze 2-arachidonoylglycerol into arachidonic acid and glycerol (Bertrand et al. 2010). One of the key features of MGL is the hydrophobic tunnel, which has been suggested to provide a model for drug research. | ||
===Metabolic Role=== | ===Metabolic Role=== | ||
===Component of Endocannabinoid System=== | ===Component of Endocannabinoid System=== | ||
Revision as of 12:01, 25 March 2014
Monoglyceride Lipase (MGL)
| |||||||||||