This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox reserved 915
From Proteopedia
(Difference between revisions)
| Line 24: | Line 24: | ||
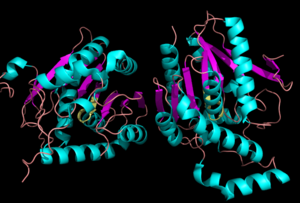
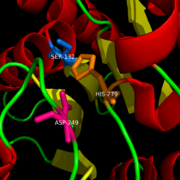
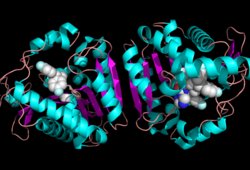
The ligand binding pocket of MGL has a large hydrophobic region with a polar bottom (Bertrand et al. 2010). Bertrand found that in MGL the binding pocket is not adjusted to the ligands shape. 2-archidonylglycerol are ligands for cannabinoid receptors (Clemente et al. 2012). Inhibition of MGL leads to increase in 2-AG levels since AG is broken down by MGL (Clemente et al. 2012). Through covalent interactions with a Cys residue, NAM, one of the many possible inhibitors, is able to inhibit MGL (Bertrand et al. 2010). <scene name='58/580298/Ligand/1'>Lignad binding site</scene> | The ligand binding pocket of MGL has a large hydrophobic region with a polar bottom (Bertrand et al. 2010). Bertrand found that in MGL the binding pocket is not adjusted to the ligands shape. 2-archidonylglycerol are ligands for cannabinoid receptors (Clemente et al. 2012). Inhibition of MGL leads to increase in 2-AG levels since AG is broken down by MGL (Clemente et al. 2012). Through covalent interactions with a Cys residue, NAM, one of the many possible inhibitors, is able to inhibit MGL (Bertrand et al. 2010). <scene name='58/580298/Ligand/1'>Lignad binding site</scene> | ||
==Overall Reaction== | ==Overall Reaction== | ||
| - | [[Image:Reaction.PNG|thumb|Overall reaction representing the hydrolysis of 2-AG by MGL]] | + | [[Image:Reaction.PNG|300px|thumb|Overall reaction representing the hydrolysis of 2-AG by MGL]] |
In this reaction 2-AG binds to the catalytic triad in the oxyanion hole in the active site. In the oxyanion hole the oxygen of the substrate is stabilized by two nitrogen atoms during the transition step of the catalytic reaction. The triad activates the nucleophilic serine and cleaves the ester bond of 2-AG that is being stabilized by its carbonyl group that is attached to the oxyanion hole. The glycerol molecule is released and it might diffuse to the narrow "exit hole", while the arachidonic acid would diffuse back to the top of the tunnel and leave the protein. | In this reaction 2-AG binds to the catalytic triad in the oxyanion hole in the active site. In the oxyanion hole the oxygen of the substrate is stabilized by two nitrogen atoms during the transition step of the catalytic reaction. The triad activates the nucleophilic serine and cleaves the ester bond of 2-AG that is being stabilized by its carbonyl group that is attached to the oxyanion hole. The glycerol molecule is released and it might diffuse to the narrow "exit hole", while the arachidonic acid would diffuse back to the top of the tunnel and leave the protein. | ||
Revision as of 13:16, 25 March 2014
Monoglyceride Lipase (MGL)
| |||||||||||