This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Daud Akhtar/Sandbox 1
From Proteopedia
(Difference between revisions)
| Line 22: | Line 22: | ||
===Substrate Binding and Catalytic Mechanism=== | ===Substrate Binding and Catalytic Mechanism=== | ||
The catalytic mechanism involved with G6PD is a general acid/base reaction. It begins with G6P binding to its active site in the second domain by interacting with Lys205. His 263 then acts as a general base that abstracts the proton from the C1-hydroxyl of G6P. Cosgrove (1998) suggest that Asp200 stabilizes the positive charge on the N atom of His264 in the transition state forming a catalytic dyad. Asp200 also plays a role in binding the phosphate moiety of G6P<ref>PMID: 9485426 </ref>. | The catalytic mechanism involved with G6PD is a general acid/base reaction. It begins with G6P binding to its active site in the second domain by interacting with Lys205. His 263 then acts as a general base that abstracts the proton from the C1-hydroxyl of G6P. Cosgrove (1998) suggest that Asp200 stabilizes the positive charge on the N atom of His264 in the transition state forming a catalytic dyad. Asp200 also plays a role in binding the phosphate moiety of G6P<ref>PMID: 9485426 </ref>. | ||
| - | [[Image:Catalytic dyad g6p.jpg|thumb| | + | [[Image:Catalytic dyad g6p.jpg|thumb|600px|Figure 2: A stick representation of the amino acid residues (Lys205, His263,Asp200) involved in the catalysis reaction associated with Glucose-6-Phosphate Dehydrogenase (PDB ID: 1QKI): CPK notation was used to depict the catalytic dyad. Image was generated using SWISSPDBViewer |
]] | ]] | ||
Revision as of 03:15, 31 March 2014
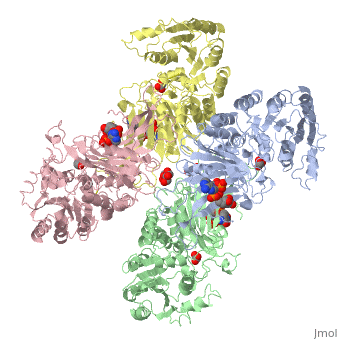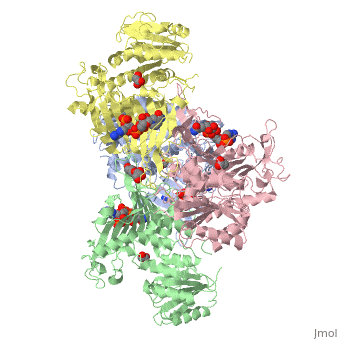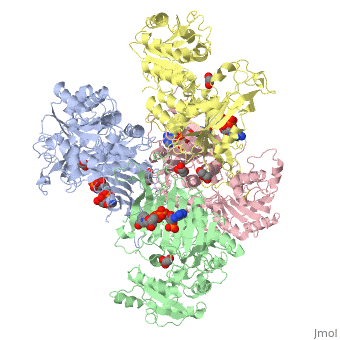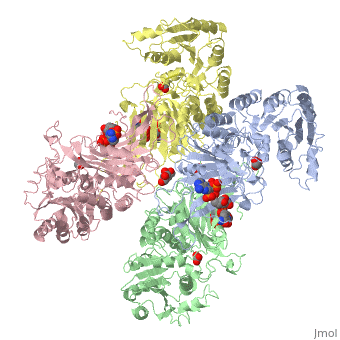
Glucose-6-Phosphate Dehydrogenase(G6PD)
| |||||||||||
References
- ↑ Salati LM, Amir-Ahmady B. Dietary regulation of expression of glucose-6-phosphate dehydrogenase. Annu Rev Nutr. 2001;21:121-40. PMID:11375432 doi:http://dx.doi.org/10.1146/annurev.nutr.21.1.121
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 2.8 2.9 Au SW, Gover S, Lam VM, Adams MJ. Human glucose-6-phosphate dehydrogenase: the crystal structure reveals a structural NADP(+) molecule and provides insights into enzyme deficiency. Structure. 2000 Mar 15;8(3):293-303. PMID:10745013
- ↑ Kotaka M, Gover S, Vandeputte-Rutten L, Au SW, Lam VM, Adams MJ. Structural studies of glucose-6-phosphate and NADP+ binding to human glucose-6-phosphate dehydrogenase. Acta Crystallogr D Biol Crystallogr. 2005 May;61(Pt 5):495-504. Epub 2005, Apr 20. PMID:15858258 doi:http://dx.doi.org/10.1107/S0907444905002350
- ↑ Corpas FJ, Barroso JB, Sandalio LM, Distefano S, Palma JM, Lupianez JA, Del Rio LA. A dehydrogenase-mediated recycling system of NADPH in plant peroxisomes. Biochem J. 1998 Mar 1;330 ( Pt 2):777-84. PMID:9480890
- ↑ Au SW, Naylor CE, Gover S, Vandeputte-Rutten L, Scopes DA, Mason PJ, Luzzatto L, Lam VM, Adams MJ. Solution of the structure of tetrameric human glucose 6-phosphate dehydrogenase by molecular replacement. Acta Crystallogr D Biol Crystallogr. 1999 Apr;55(Pt 4):826-34. PMID:10089300
- ↑ Bhadbhade MM, Adams MJ, Flynn TG, Levy HR. Sequence identity between a lysine-containing peptide from Leuconostoc mesenteroides glucose-6-phosphate dehydrogenase and an active site peptide from human erythrocyte glucose-6-phosphate dehydrogenase. FEBS Lett. 1987 Jan 26;211(2):243-6. PMID:3100332
- ↑ Cosgrove MS, Naylor C, Paludan S, Adams MJ, Levy HR. On the mechanism of the reaction catalyzed by glucose 6-phosphate dehydrogenase. Biochemistry. 1998 Mar 3;37(9):2759-67. PMID:9485426 doi:10.1021/bi972069y
- ↑ Ramos KL, Colquhoun A. Protective role of glucose-6-phosphate dehydrogenase activity in the metabolic response of C6 rat glioma cells to polyunsaturated fatty acid exposure. Glia. 2003 Aug;43(2):149-66. PMID:12838507 doi:http://dx.doi.org/10.1002/glia.10246
- ↑ Tian WN, Braunstein LD, Pang J, Stuhlmeier KM, Xi QC, Tian X, Stanton RC. Importance of glucose-6-phosphate dehydrogenase activity for cell growth. J Biol Chem. 1998 Apr 24;273(17):10609-17. PMID:9553122
- ↑ Scott MD, Zuo L, Lubin BH, Chiu DT. NADPH, not glutathione, status modulates oxidant sensitivity in normal and glucose-6-phosphate dehydrogenase-deficient erythrocytes. Blood. 1991 May 1;77(9):2059-64. PMID:2018843
- ↑ Scott MD, Zuo L, Lubin BH, Chiu DT. NADPH, not glutathione, status modulates oxidant sensitivity in normal and glucose-6-phosphate dehydrogenase-deficient erythrocytes. Blood. 1991 May 1;77(9):2059-64. PMID:2018843
- ↑ . Glucose-6-phosphate dehydrogenase deficiency. WHO Working Group. Bull World Health Organ. 1989;67(6):601-11. PMID:2633878
- ↑ Manganelli G, Masullo U, Passarelli S, Filosa S. Glucose-6-phosphate dehydrogenase deficiency: disadvantages and possible benefits. Cardiovasc Hematol Disord Drug Targets. 2013 Mar 1;13(1):73-82. PMID:23534950
- ↑ Beutler E. Glucose-6-phosphate dehydrogenase deficiency. N Engl J Med. 1991 Jan 17;324(3):169-74. PMID:1984194 doi:http://dx.doi.org/10.1056/NEJM199101173240306