This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox reserved 915
From Proteopedia
(Difference between revisions)
| Line 3: | Line 3: | ||
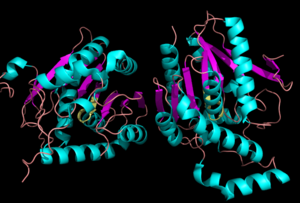
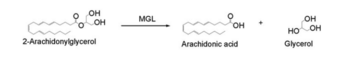
[[Image:Complete_crystal_structure.png|left|300px|thumb|'''Figure 1:'''Crystal Structure of MGL Alpha helixes are in blue and beta sheets in purple. This protein is a dimer that is linked by antiparallel beta sheets]] | [[Image:Complete_crystal_structure.png|left|300px|thumb|'''Figure 1:'''Crystal Structure of MGL Alpha helixes are in blue and beta sheets in purple. This protein is a dimer that is linked by antiparallel beta sheets]] | ||
==Background== | ==Background== | ||
| - | Monoglyceride [[:Category:Lipase| Lipase]] (MGL) is part of the α/β hydrolase family,a [[:Category:Serine hydrolase| Serine hydrolase]] (Figure 1), having a <scene name='58/580298/Catalytic_triad/4'>Ser-His-Asp catalytic triad </scene> <ref name="Clemente">[Clemente, J. C., E. Nulton, M. Nelen, M. J. Todd, D. Maguire, C. Schalk-Hihi, L. C. Kuo, S.-P. Zhang, C. M. Flores, and J. K. Kranz. "Screening and Characterization of Human Monoglyceride Lipase Active Site Inhibitors Using Orthogonal Binding and Functional Assays." Journal of Biomolecular Screening 17.5 (2012): 629-40]</ref>. [http://en.wikipedia.org/wiki/Monoacylglycerol_lipase MGL] is present in most cells, providing the rate limiting step for the hydrolysis of [http://en.wikipedia.org/wiki/Monoglyceride monoacylglycerols] (MG) into fatty acids and glycerol <ref name="Taschler">[Taschler, U., F. P. W. Radner, C. Heier, R. Schreiber, M. Schweiger, G. Schoiswohl, K. Preiss-Landl, D. Jaeger, B. Reiter, H. C. Koefeler, J. Wojciechowski, C. Theussl, J. M. Penninger, A. Lass, G. Haemmerle, R. Zechner, and R. Zimmermann. "Monoglyceride Lipase Deficiency in Mice Impairs Lipolysis and Attenuates Diet-induced Insulin Resistance." Journal of Biological Chemistry 286.20 (2011): 17467-7477]</ref> . MGL also terminates the signaling of a primary endocannabinoid, 2-arachidonoyl glycerol (2-AG) <ref name="Savinainen">[Savinainen, Juha R., Megumi Yoshino, Anna Minkkilä, Tapio Nevalainen, and Jarmo T. Laitinen. "Characterization of Binding Properties of Monoglyceride Lipase Inhibitors by a Versatile Fluorescence-based Technique." Analytical Biochemistry 399.1 (2010): 132-34]</ref>. MGL is the main enzyme responsible for hydrolyzing 2-arachidonoylglycerol into arachidonic acid and glycerol ''in vivo'' (Figure 3) <ref name="Bertrand">[ Bertrand, T., F. Augé, J. Houtmann, A. Rak, F. Vallée, V. Mikol, P.f. Berne, N. Michot, D. Cheuret, C. Hoornaert, and M. Mathieu. "Structural Basis for Human Monoglyceride Lipase Inhibition." Journal of Molecular Biology 396.3 (2010): 663-73.]</ref>. One of the key features of MGL is the hydrophobic tunnel, which has been suggested to provide a model for drug research (Figure 7). | + | Monoglyceride [[:Category:Lipase| Lipase]] (MGL) is part of the α/β hydrolase family,a [[:Category:Serine hydrolase| Serine hydrolase]] (Figure 1), having a <scene name='58/580298/Catalytic_triad/4'>Ser-His-Asp catalytic triad </scene> <ref name="Clemente">[Clemente, J. C., E. Nulton, M. Nelen, M. J. Todd, D. Maguire, C. Schalk-Hihi, L. C. Kuo, S.-P. Zhang, C. M. Flores, and J. K. Kranz. "Screening and Characterization of Human Monoglyceride Lipase Active Site Inhibitors Using Orthogonal Binding and Functional Assays." Journal of Biomolecular Screening 17.5 (2012): 629-40]</ref>. [http://en.wikipedia.org/wiki/Monoacylglycerol_lipase MGL] is present in most cells, providing the rate limiting step for the hydrolysis of [http://en.wikipedia.org/wiki/Monoglyceride monoacylglycerols] (MG) into fatty acids and glycerol <ref name="Taschler">[Taschler, U., F. P. W. Radner, C. Heier, R. Schreiber, M. Schweiger, G. Schoiswohl, K. Preiss-Landl, D. Jaeger, B. Reiter, H. C. Koefeler, J. Wojciechowski, C. Theussl, J. M. Penninger, A. Lass, G. Haemmerle, R. Zechner, and R. Zimmermann. "Monoglyceride Lipase Deficiency in Mice Impairs Lipolysis and Attenuates Diet-induced Insulin Resistance." Journal of Biological Chemistry 286.20 (2011): 17467-7477]</ref> . MGL also terminates the signaling of a primary endocannabinoid, 2-arachidonoyl glycerol (2-AG) <ref name="Savinainen">[Savinainen, Juha R., Megumi Yoshino, Anna Minkkilä, Tapio Nevalainen, and Jarmo T. Laitinen. "Characterization of Binding Properties of Monoglyceride Lipase Inhibitors by a Versatile Fluorescence-based Technique." Analytical Biochemistry 399.1 (2010): 132-34]</ref>. MGL is the main enzyme responsible for hydrolyzing 2-arachidonoylglycerol into arachidonic acid and glycerol ''in vivo'' (Figure 3) <ref name="Bertrand">[ Bertrand, T., F. Augé, J. Houtmann, A. Rak, F. Vallée, V. Mikol, P.f. Berne, N. Michot, D. Cheuret, C. Hoornaert, and M. Mathieu. "Structural Basis for Human Monoglyceride Lipase Inhibition." Journal of Molecular Biology 396.3 (2010): 663-73.]</ref>. One of the key features of MGL is the hydrophobic tunnel, which has been suggested to provide a model for drug research (Figure 7). <ref name="Bertrand" /> |
===Metabolic Role=== | ===Metabolic Role=== | ||
| Line 28: | Line 28: | ||
==Structure== | ==Structure== | ||
| - | The <scene name='58/580298/Overall_structure/3'>overall structure</scene> of MGL has eight-stranded β-sheet protein fold with seven parallel and one <scene name='58/580299/Beta_sheets/1'> antiparallel strand </scene>. Similar to the other α/β hydrolases, the β-sheets in the center of the protein surrounded by α-helices. The combination of the α-helices and β-sheets are able to provide a stable scaffold for the active site within MGL. Within the main domain of MGL is the conserved catalytic triad <ref name="Bertrand" />. | + | The <scene name='58/580298/Overall_structure/3'>overall structure</scene> of MGL has an eight-stranded β-sheet protein fold with seven parallel and one <scene name='58/580299/Beta_sheets/1'> antiparallel strand </scene>. Similar to the other α/β hydrolases, the β-sheets in the center of the protein surrounded by α-helices. The combination of the α-helices and β-sheets are able to provide a stable scaffold for the active site within MGL. Within the main domain of MGL is the conserved catalytic triad <ref name="Bertrand" />. |
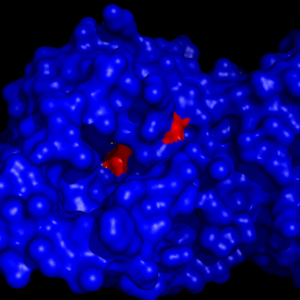
== Catalytic triad == | == Catalytic triad == | ||
Revision as of 19:13, 19 April 2014
Monoglyceride Lipase (MGL)
| |||||||||||