You may include any references to papers as in: the use of JSmol in Proteopedia [1] or to the article describing Jmol [2] to the rescue.
Intro
Erythropoietin (EPO) is a hormone produced in the kidneys that stimulates the formation of red blood cells. EPO is a glycoprotein that is stimulated when the levels of O2 are abnormally low. This event signals more red blood cells to made from the erythrocytes. Abnormal levels of erythropoietin can be associated with bone marrow disorders, kidney disease, or a synthesized recombinant form that has been injected into the blood stream. Synthesized recombinant EPO has made many headlines in the past few years, due to its use to by Tour de France athletes. They used EPO to illegally dope their blood and increase the amount of oxygen that can be consumed by the body at the time of administration thus increasing endurance. They used EPO because there was no test at the time that could differentiate between naturally produced EPO and the form that was injected.
History
Miyake et al first purified a few milligrams of erythropoietin from 2500 L human urine in 1977 [3]. This glycoprotein was first suspected as doctors observed anemia patients. Researchers noticed that patients with anemia had increased levels of erythropoiesis [4]. They noticed that the level of oxygen, seemed to be correlated with the amount of erythropoiesis, as well. After isolation, researchers worked to clone the gene so that they could produce the glycoprotein and treat hypoxia. The FDA approved this type of treatment in the 1990’s [5]. EPO works to stimulate the amount of red blood cells and the efficiency of hemoglobin, which increases oxygen library. This trait is what has led to many athletes to abuse this glycoprotein to improve their performance in their athletic sport, the most famous being Tour de France competitors (8). Most athletes involved in these scandal would intravenously inject themselves with this hormone (7). However, as medical techniques have advanced, some have started gene doping through in vivo and ex vivo gene transfer (7).
Structure.
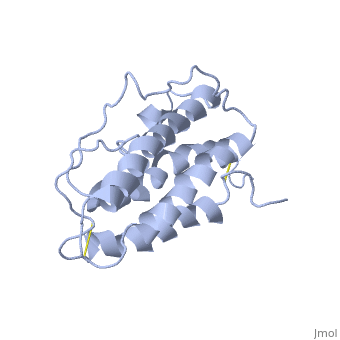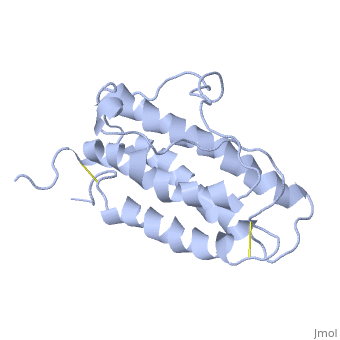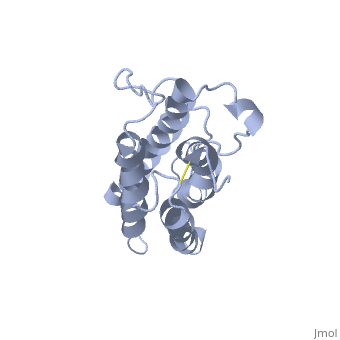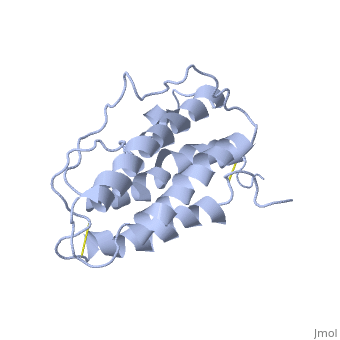
The gene for EPO is found on chromosome 7 and is composed of five exons and four introns [6]. The transcriptional product of this gene is an amino acid chain of 193 bases. During translation, the chain is modified to 166 amino acids [7]. The cleaved 27 amino acid leader sequence is made mostly of hydrophobic amino acids. After translation is complete, the C-terminus loses its final arginine residue to reach its final length of 165 amino acids residues [8]. The total glycoprotein weighs 30 kDa with the peptide backbone weighing 18 kDa. EPO is a glycoprotein composed of only [9]. The sulfur of the cysteine residues links to form disulfide bonds. These disulfide bonds help keep EPO's structure. Helix A is connected to Helix D by , while Helix A and Helix B are connected by . EPO’s structure was determined in 1993. It is made up of four alpha helixes. EPO is produced mainly in the kidney, but further research has shown the brain and liver still produce small amounts [10].
Stimulus
The levels of oxygen found in the blood moderate the process of erythropoiesis, which is the production of red blood cells. At low levels of oxygen, EPO released into the blood stream from the kidneys in adults humans. After release, the hormone travels to the bone marrow and binds to receptors, which starts the proliferation of red blood cells to increase the oxygen consumption. This increase in consumption will hopefully reestablish normal levels of blood in the system.
Receptors
The of the blood marrow is part of the hematipoietic cytokine family. This receptor has a single transmembrane domain, that forms a homodimer complex until it is activated by the binding of EPO.This receptor is 484 amino acids long and weigh 52.6 kDa [11]. Once the homodimer is formed after the binding, autophosphorlation of the Jak2 kinases, which activates other cellular processes [12]. This transmembrane receptor has two extracellular domains. This receptor has two disulfide bonds that are formed from 4 cystine residues, [13]. The intracellular domain of this receptor does not possess any enzymatic activity like other receptors. When EPO comes in contact with the extracellular domains form a ligand bond. The extracellular sinding site 1 and Binding site 2 are composed of [14]. When EPO binds, all loops on D1 and D2 of binding site one form a bind with EPO. However loop 4 of D1 on binding site 2 does not participate in the binding of EPO [15]. After the biniding of EPO, 8 tyrosine residues are phosphoralated which activates the [16]. This kinase helps regulate the transcription of different genes and expression of other proteins.
References
- ↑ doi: https://dx.doi.org/10.1007/BF02934992
- ↑ Syed RS, Reid SW, Li C, Cheetham JC, Aoki KH, Liu B, Zhan H, Osslund TD, Chirino AJ, Zhang J, Finer-Moore J, Elliott S, Sitney K, Katz BA, Matthews DJ, Wendoloski JJ, Egrie J, Stroud RM. Efficiency of signalling through cytokine receptors depends critically on receptor orientation. Nature. 1998 Oct 1;395(6701):511-6. PMID:9774108 doi:http://dx.doi.org/10.1038/26773
- ↑ Kawakita M, Ogawa M, Goldwasser E, Miyake T. Characterization of human megakaryocyte colony-stimulating factor in the urinary extracts from patients with aplastic anemia and idiopathic thrombocytopenic purpura. Blood. 1983 Mar;61(3):556-60. PMID:6600633
- ↑ Jelkmann W. Erythropoietin after a century of research: younger than ever. Eur J Haematol. 2007 Mar;78(3):183-205. Epub 2007 Jan 23. PMID:17253966 doi:http://dx.doi.org/10.1111/j.1600-0609.2007.00818.x
- ↑ Jelkmann W. Erythropoietin after a century of research: younger than ever. Eur J Haematol. 2007 Mar;78(3):183-205. Epub 2007 Jan 23. PMID:17253966 doi:http://dx.doi.org/10.1111/j.1600-0609.2007.00818.x
- ↑ Jelkmann W. Erythropoietin: structure, control of production, and function. Physiol Rev. 1992 Apr;72(2):449-89. PMID:1557429
- ↑ Jelkmann W. Erythropoietin after a century of research: younger than ever. Eur J Haematol. 2007 Mar;78(3):183-205. Epub 2007 Jan 23. PMID:17253966 doi:http://dx.doi.org/10.1111/j.1600-0609.2007.00818.x
- ↑ Erslev, A. J., and J. Caro. "Physiologic and molecular biology of erythropoietin." Medical oncology and tumor pharmacotherapy 3.3-4 (1986): 159-164.
- ↑ Erslev, A. J., and J. Caro. "Physiologic and molecular biology of erythropoietin." Medical oncology and tumor pharmacotherapy 3.3-4 (1986): 159-164.
- ↑ Erslev, A. J., and J. Caro. "Physiologic and molecular biology of erythropoietin." Medical oncology and tumor pharmacotherapy 3.3-4 (1986): 159-164.
- ↑ Jelkmann W. Erythropoietin after a century of research: younger than ever. Eur J Haematol. 2007 Mar;78(3):183-205. Epub 2007 Jan 23. PMID:17253966 doi:http://dx.doi.org/10.1111/j.1600-0609.2007.00818.x
- ↑ doi: https://dx.doi.org/10.1016/S1043-2760(98)00101-5
- ↑ doi: https://dx.doi.org/10.1016/S1043-2760(98)00101-5
- ↑ doi: https://dx.doi.org/10.1016/S1043-2760(98)00101-5
- ↑ Syed RS, Reid SW, Li C, Cheetham JC, Aoki KH, Liu B, Zhan H, Osslund TD, Chirino AJ, Zhang J, Finer-Moore J, Elliott S, Sitney K, Katz BA, Matthews DJ, Wendoloski JJ, Egrie J, Stroud RM. Efficiency of signalling through cytokine receptors depends critically on receptor orientation. Nature. 1998 Oct 1;395(6701):511-6. PMID:9774108 doi:http://dx.doi.org/10.1038/26773
- ↑ doi: https://dx.doi.org/10.1016/S1043-2760(98)00101-5
1. Constantinescu, Stefan N., Saghi Ghaffar, Harvey F. Lodish. The Erythropoietin Receptor: Structure, Activation and Intracellular Signal Transduction. Trends in Endocrinology
and Metabolism, Volume10, Issue 1, 1January 1999, Pages 18-23.
2. Erslev, A. J., and J. Caro. "Physiologic and molecular biology of erythropoietin." Medical oncology and tumor pharmacotherapy 3.3-4 (1986): 159-164.
3.Jelkmann, Wolfgang. Erythropoietin after a century of research: younger than ever. European Journal of Haematology. Volume 78, Issue 3, 23 Jan 2007 , Pages 183-205.
4. Jelkmann W: Erythropoietin: Structure, control of production, and function. Physiol Rev 72:449–489, 1992
5.Kawakita, Makoto, et al. "Characterization of human megakaryocyte colony-stimulating factor in the urinary extracts from patients with aplastic anemia and idiopathic thrombocytopenic purpura." Blood 61.3 (1983): 556-560.
6. Syed, Rashid S., Reid, Scott W., Li, Cuiwei. Efficiency Signaling Through Cytokine receptors Depends Critically on Receptor Orientation. Nature 395, 511-516, 1 October 1998.
7. http://www.bbc.com/news/magazine-25687002
8. http://news.bbc.co.uk/sport2/hi/front_page/4657008.stm