This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 186
From Proteopedia
(Difference between revisions)
| Line 8: | Line 8: | ||
== Secondary Structure and the Thioredoxin Like Fold == | == Secondary Structure and the Thioredoxin Like Fold == | ||
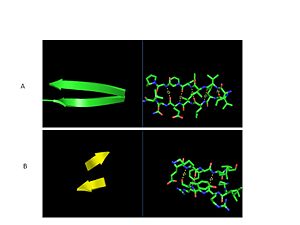
| - | GPx-1 consists of nine β-strand nine α-helices with four of the helices being of the 310 form (Figure 2). Interestingly two of the β-strands form a parallel β-sheet (Figure 3)[[Image:Beta_sheets3.jpg|300px| | + | GPx-1 consists of nine β-strand nine α-helices with four of the helices being of the 310 form (Figure 2). Interestingly two of the β-strands form a parallel β-sheet (Figure 3)[[Image:Beta_sheets3.jpg|300px|right|thumb|]]. The classic thioredoxin fold consists of a four stranded β-sheet that is surrounded by three α-helices (5). However the thioredoxin fold is commonly subject to the insertion of additional secondary structural elements between the second β-strand and the second α-helices (6). This is seen in GPx-1 as there is an addition of an α-helix and a β-strand between the second β-strand and the second α-helices (6). A similar insertion is found in peroxiredoxins, a different family of proteins which also catalyze the reduction of hydroperoxides (6). |
== Relevance == | == Relevance == | ||
Revision as of 05:05, 30 April 2014
| |||||||||||