This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 186
From Proteopedia
(Difference between revisions)
| Line 7: | Line 7: | ||
Glutathione peroxidase 1 (GPx-1) is a tetramer (23 kDa per monomer) with two units composed of dimers. GPx-1 is the most abundant member of the Glutathione peroxidase family. It is found in all cells and is located in the cytosolic and mitochondrial compartments (1). GPx-1 is a crucial anti-oxidant enzyme that catalyzes the conversion of hydrogen peroxide into water (2). Interestingly GPx-1 contains the rare amino acid selenocysteine which acts as the peroxidatic residue (2). The overall reaction that GPx-1 catalyzes is H2O2 + 2Glutathione (GSH) -> 2H20 + GS-SG (Figure 1). In addition to hydrogen peroxide GPx-1 can reduce other soluble hydroperoxides including lipid hydroperoxides (3). Because of its role in regulating the intracellular concentration of reactive oxygen species, GPx-1 has been found to play a role in numerous processes including cell proliferation, apoptosis, and inflammation (1). Furthermore deficiencies in GPx-1 has been linked to the development of cancers, neurodegenerative diseases, and heart disease (4). | Glutathione peroxidase 1 (GPx-1) is a tetramer (23 kDa per monomer) with two units composed of dimers. GPx-1 is the most abundant member of the Glutathione peroxidase family. It is found in all cells and is located in the cytosolic and mitochondrial compartments (1). GPx-1 is a crucial anti-oxidant enzyme that catalyzes the conversion of hydrogen peroxide into water (2). Interestingly GPx-1 contains the rare amino acid selenocysteine which acts as the peroxidatic residue (2). The overall reaction that GPx-1 catalyzes is H2O2 + 2Glutathione (GSH) -> 2H20 + GS-SG (Figure 1). In addition to hydrogen peroxide GPx-1 can reduce other soluble hydroperoxides including lipid hydroperoxides (3). Because of its role in regulating the intracellular concentration of reactive oxygen species, GPx-1 has been found to play a role in numerous processes including cell proliferation, apoptosis, and inflammation (1). Furthermore deficiencies in GPx-1 has been linked to the development of cancers, neurodegenerative diseases, and heart disease (4). | ||
| - | == Secondary Structure and the Thioredoxin Like Fold == | + | == Secondary Structure and the Thioredoxin Like Fold of GPx-1 == |
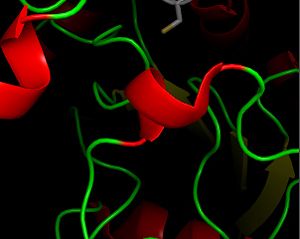
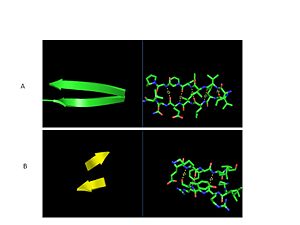
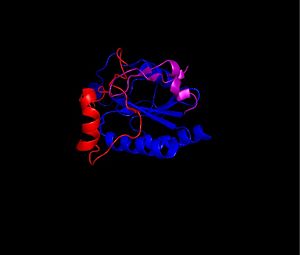
| - | The secondary structure of GPx-1 consists of nine β-strands and nine α-helices with five of the helices being of the 310 form (Figure 2)[[Image:Helix310.jpg|300px|left|thumb|]]. Interestingly two of the β-strands form a parallel β-sheet (Figure 3)[[Image:Beta_sheets3.jpg|300px|right|thumb|]]. GPx-1 exhibits a thioredoxin like fold. The classic thioredoxin fold consists of a four stranded β-sheet that is surrounded by three α-helices (5). However the thioredoxin fold is commonly subject to the insertion of additional secondary structural elements between the second β-strand and the second α-helices (6). This is seen in GPx-1 as there is an addition of an α-helix and a β-strand between the second β-strand and the second α-helices (6). A similar insertion is found in peroxiredoxins, a different family of proteins which also catalyze the reduction of hydroperoxides(6). | + | The secondary structure of GPx-1 consists of nine β-strands and nine α-helices with five of the helices being of the 310 form (Figure 2)[[Image:Helix310.jpg|300px|left|thumb|]]. Interestingly two of the β-strands form a parallel β-sheet (Figure 3)[[Image:Beta_sheets3.jpg|300px|right|thumb|]]. GPx-1 exhibits a thioredoxin like fold. The classic thioredoxin fold consists of a four stranded β-sheet that is surrounded by three α-helices (Figure 4) (5). However the thioredoxin fold is commonly subject to the insertion of additional secondary structural elements between the second β-strand and the second α-helices (6). This is seen in GPx-1 as there is an addition of an α-helix and a β-strand between the second β-strand and the second α-helices (Figure 5)[[Image:Thioredoxin_like_fold.jpg|300px|right|thumb|]] (6). A similar insertion is found in peroxiredoxins, a different family of proteins which also catalyze the reduction of hydroperoxides(6). |
== Relevance == | == Relevance == | ||
Revision as of 06:47, 30 April 2014
| |||||||||||