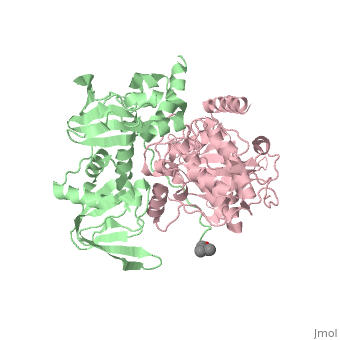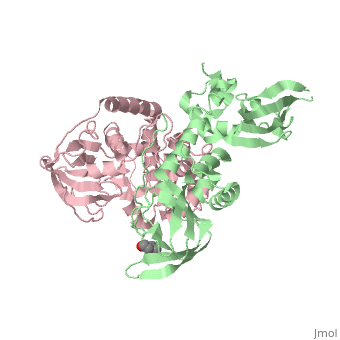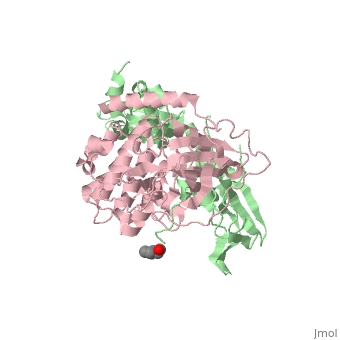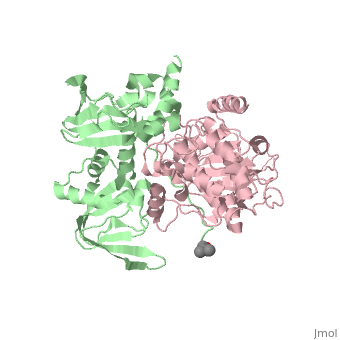This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Protein Kinase A
From Proteopedia
| Line 7: | Line 7: | ||
== Structure == | == Structure == | ||
| - | PKA consists of two types of subunits. It has a 49-kd regulatory <scene name='58/582909/Regulatory_subunits/1'>regulatory subunit</scene> (R) and a 38-kd <scene name='58/582909/Catalytic_subunits/1'>catalytic subunit</scene> (C). The R dimer contains two cAMP bindng domains and the pseudosbustrate sequence, Arg-Arg-Gly-Ala-Ile. The C subunit has two lobes. The larger lobe binds the peptide and contributes the key catalytic residues and the smaller lobe binds ATP-Mg2+. This <scene name='58/582909/Catalytic_subunit_1/1'>scene</scene> shows a catalytic subunit with an | + | PKA consists of two types of subunits. It has a 49-kd regulatory <scene name='58/582909/Regulatory_subunits/1'>regulatory subunit</scene> (R) and a 38-kd <scene name='58/582909/Catalytic_subunits/1'>catalytic subunit</scene> (C). The R dimer contains two cAMP bindng domains and the pseudosbustrate sequence, Arg-Arg-Gly-Ala-Ile. The C subunit has two lobes. The larger lobe binds the peptide and contributes the key catalytic residues and the smaller lobe binds ATP-Mg2+. This <scene name='58/582909/Catalytic_subunit_1/1'>scene</scene> shows a catalytic subunit with an inhibitor with a psuedosequence bound to the active site and an ATP bound to the adjacent active site. When PKA is enzymatically inactive (absence of cAMP), it is a heterotetrameric complex of a regulatory dimer bound to two catalytic subunits, forming the R2C2 complex. In its active state, the complex dissociates to form an R2 subunit and two C subunits. The C subunits are enzymatically active in itself and once released, are free to bind and phosphorylate available substrate proteins. |
== Mechanism == | == Mechanism == | ||
Revision as of 17:19, 30 April 2014
Protein kinase A (PKA)
| |||||||||||
References
[1]Herraez A. Biomolecules in the computer: Jmol to the rescue. Biochem Mol Biol Educ. 2006 Jul;34(4):255-61. doi: 10.1002/bmb.2006.494034042644. PMID:21638687 doi:10.1002/bmb.2006.494034042644 [2]C. Kim, N.-H. Xuong, and S.S. Taylor, "Crystal structure of a complex between catalytic and regulatory (Rlα) subunits of PKA,"Science 307, 690 (2005) doi:10.2210/rcsb_pdb/mom_2012_8