This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Raghad zoubi
From Proteopedia
(Difference between revisions)
| Line 2: | Line 2: | ||
<StructureSection load='1hvx' size='340' side='right' caption='α-Ameylase' scene=''> | <StructureSection load='1hvx' size='340' side='right' caption='α-Ameylase' scene=''> | ||
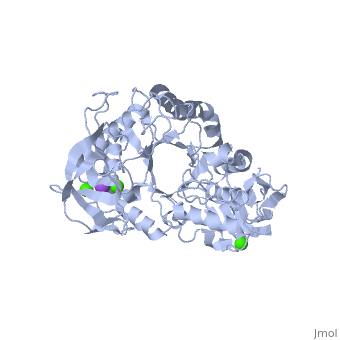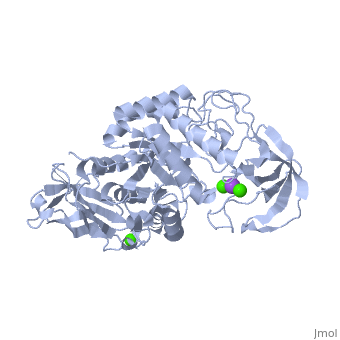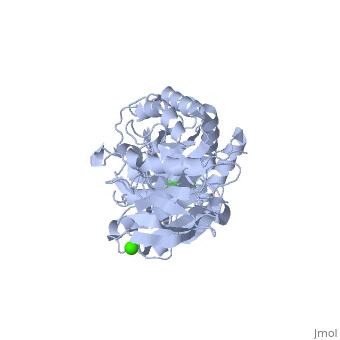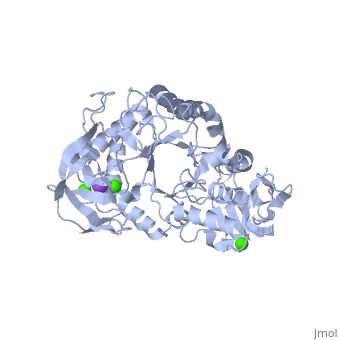
| + | α-Amylase is a protein enzyme EC 3.2.1.1 that hydrolyses alpha bonds of large, alpha-linked polysaccharides, such as starch and glycogen, yielding glucose and maltose. It is the major form of amylase found in humans and other mammals.It is also present in seeds containing starch as a food reserve, and is secreted by many fungi. <ref>[http://en.wikipedia.org/wiki/Alpha-amylase]</ref> | ||
== Introduction == | == Introduction == | ||
The <scene name='60/609816/Alpha-amylase/1'>Alpha-amylase</scene> (EC 3.2.1.1 ) (CAS# 9014-71-5) (alternative names: 1,4-α-D-glucan glucanohydrolase; glycogenase) are calcium metalloenzymes, completely unable to function in the absence of calcium. By acting at random locations along the starch chain, α-amylase breaks down long-chain carbohydrates, ultimately yielding maltotriose and maltose from amylose, or maltose, glucose and "limit dextrin" from amylopectin. Because it can act anywhere on the substrate, α-amylase tends to be faster-acting than β-amylase. In animals, it is a major digestive enzyme, and its optimum pH is 6.7–7.0 | The <scene name='60/609816/Alpha-amylase/1'>Alpha-amylase</scene> (EC 3.2.1.1 ) (CAS# 9014-71-5) (alternative names: 1,4-α-D-glucan glucanohydrolase; glycogenase) are calcium metalloenzymes, completely unable to function in the absence of calcium. By acting at random locations along the starch chain, α-amylase breaks down long-chain carbohydrates, ultimately yielding maltotriose and maltose from amylose, or maltose, glucose and "limit dextrin" from amylopectin. Because it can act anywhere on the substrate, α-amylase tends to be faster-acting than β-amylase. In animals, it is a major digestive enzyme, and its optimum pH is 6.7–7.0 | ||
Revision as of 16:11, 22 November 2014
Example page for α-Ameylase
| |||||||||||