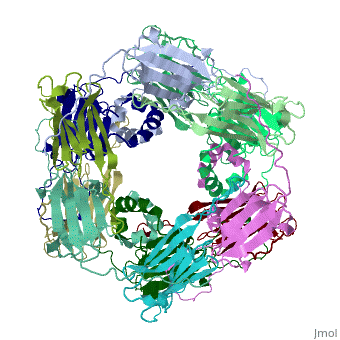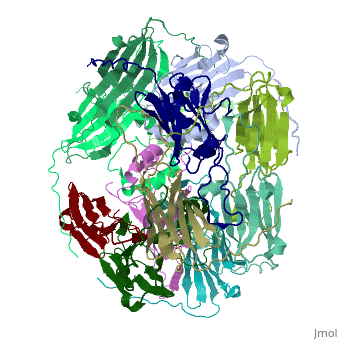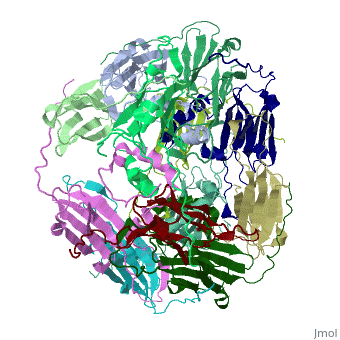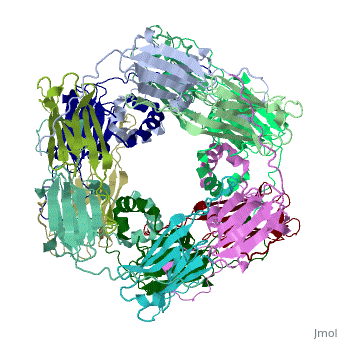This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Keithballard/sandbox
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
==Structure of Class I cytosolic sHSP== | ==Structure of Class I cytosolic sHSP== | ||
| - | <StructureSection load='1gme' size='340' side='right' caption='Caption for this structure' scene=''> | + | <StructureSection load='1gme' size='340' side='right' caption='Caption for this structure' scene=''><scene name='60/609774/Dimer_default/2'></scene> |
==Introduction== | ==Introduction== | ||
Small heat shock proteins (sHSPs) and related α-crystallins are virtually ubiquitous, ATP-independent molecular chaperones linked to diseases of protein misfolding. They comprise a conserved core α-crystallin domain (ACD) flanked by an evolutionarily variable N-terminal arm (NTA) and semi-conserved C-terminal extension. They are capable of binding up to an equal mass of unfolding protein, forming large, heterogeneous sHSP-substrate complexes that make substrate available to the ATP-dependent chaperones for refolding. | Small heat shock proteins (sHSPs) and related α-crystallins are virtually ubiquitous, ATP-independent molecular chaperones linked to diseases of protein misfolding. They comprise a conserved core α-crystallin domain (ACD) flanked by an evolutionarily variable N-terminal arm (NTA) and semi-conserved C-terminal extension. They are capable of binding up to an equal mass of unfolding protein, forming large, heterogeneous sHSP-substrate complexes that make substrate available to the ATP-dependent chaperones for refolding. | ||
Revision as of 14:48, 3 December 2014
Structure of Class I cytosolic sHSP
| |||||||||||