This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Erythropoietin
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
==Erythropoietin Structure, Function, and History== | ==Erythropoietin Structure, Function, and History== | ||
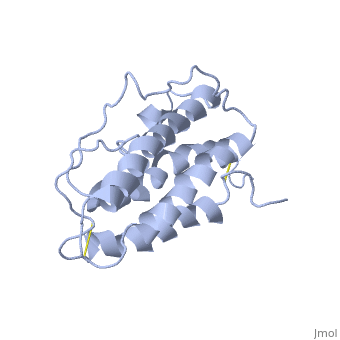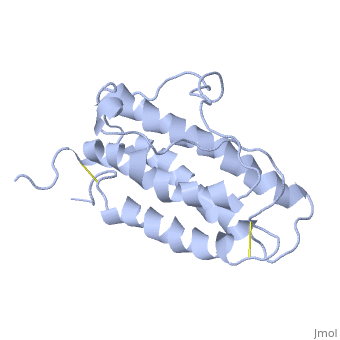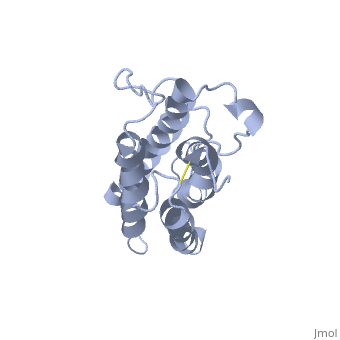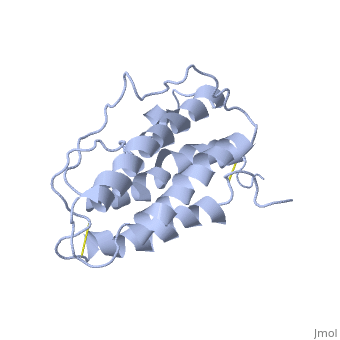
| - | <StructureSection load='1buy' size=' | + | <StructureSection load='1buy' size='350' side='right' caption='Human erythropoietin NMR structure (PDB code [[1buy]]).' scene=> |
==Introduction== | ==Introduction== | ||
| Line 34: | Line 34: | ||
and Metabolism, Volume10, Issue 1, 1January 1999, Pages 18-23.</ref>. The intracellular domain of this receptor does not possess any enzymatic activity like other receptors. When EPO comes in contact with the extracellular domains form a ligand bond. The extracellular sinding site 1 and Binding site 2 are composed of <scene name='58/583377/Eporeceptord1d2/1'>D1 and D2</scene> <ref>Constantinescu, Stefan N., Saghi Ghaffar, Harvey F. Lodish. The Erythropoietin Receptor: Structure, Activation and Intracellular Signal Transduction. Trends in Endocrinology and Metabolism, Volume10, Issue 1, 1January 1999, Pages 18-23.</ref>. When EPO binds, all loops on D1 and D2 of binding site one form a bind with EPO. However loop 4 of D1 on binding site 2 does not participate in the binding of EPO <ref>PMID: 9774108</ref>. After the biniding of EPO, 8 tyrosine residues are phosphoralated which activates the <scene name='58/583377/Jak2/2'>Jak2 kinase</scene> <ref>Constantinescu, Stefan N., Saghi Ghaffar, Harvey F. Lodish. The Erythropoietin Receptor: Structure, Activation and Intracellular Signal Transduction. Trends in Endocrinology | and Metabolism, Volume10, Issue 1, 1January 1999, Pages 18-23.</ref>. The intracellular domain of this receptor does not possess any enzymatic activity like other receptors. When EPO comes in contact with the extracellular domains form a ligand bond. The extracellular sinding site 1 and Binding site 2 are composed of <scene name='58/583377/Eporeceptord1d2/1'>D1 and D2</scene> <ref>Constantinescu, Stefan N., Saghi Ghaffar, Harvey F. Lodish. The Erythropoietin Receptor: Structure, Activation and Intracellular Signal Transduction. Trends in Endocrinology and Metabolism, Volume10, Issue 1, 1January 1999, Pages 18-23.</ref>. When EPO binds, all loops on D1 and D2 of binding site one form a bind with EPO. However loop 4 of D1 on binding site 2 does not participate in the binding of EPO <ref>PMID: 9774108</ref>. After the biniding of EPO, 8 tyrosine residues are phosphoralated which activates the <scene name='58/583377/Jak2/2'>Jak2 kinase</scene> <ref>Constantinescu, Stefan N., Saghi Ghaffar, Harvey F. Lodish. The Erythropoietin Receptor: Structure, Activation and Intracellular Signal Transduction. Trends in Endocrinology | ||
and Metabolism, Volume10, Issue 1, 1January 1999, Pages 18-23.</ref>. This kinase helps regulate the transcription of different genes and expression of other proteins. | and Metabolism, Volume10, Issue 1, 1January 1999, Pages 18-23.</ref>. This kinase helps regulate the transcription of different genes and expression of other proteins. | ||
| + | </StructureSection> | ||
==3D structures of erythropoietin== | ==3D structures of erythropoietin== | ||
Revision as of 14:33, 20 January 2016
Erythropoietin Structure, Function, and History
| |||||||||||
3D structures of erythropoietin
Updated on 20-January-2016
1cn4 – hEP (mutant) + EP receptor – human
1buy – hEP (mutant) - NMR
1eer – hEP (mutant) + EP receptor (mutant)
References
- ↑ Kawakita M, Ogawa M, Goldwasser E, Miyake T. Characterization of human megakaryocyte colony-stimulating factor in the urinary extracts from patients with aplastic anemia and idiopathic thrombocytopenic purpura. Blood. 1983 Mar;61(3):556-60. PMID:6600633
- ↑ Jelkmann W. Erythropoietin after a century of research: younger than ever. Eur J Haematol. 2007 Mar;78(3):183-205. Epub 2007 Jan 23. PMID:17253966 doi:http://dx.doi.org/10.1111/j.1600-0609.2007.00818.x
- ↑ Jelkmann W. Erythropoietin after a century of research: younger than ever. Eur J Haematol. 2007 Mar;78(3):183-205. Epub 2007 Jan 23. PMID:17253966 doi:http://dx.doi.org/10.1111/j.1600-0609.2007.00818.x
- ↑ Jelkmann W. Erythropoietin: structure, control of production, and function. Physiol Rev. 1992 Apr;72(2):449-89. PMID:1557429
- ↑ Jelkmann W. Erythropoietin after a century of research: younger than ever. Eur J Haematol. 2007 Mar;78(3):183-205. Epub 2007 Jan 23. PMID:17253966 doi:http://dx.doi.org/10.1111/j.1600-0609.2007.00818.x
- ↑ Erslev, A. J., and J. Caro. "Physiologic and molecular biology of erythropoietin." Medical oncology and tumor pharmacotherapy 3.3-4 (1986): 159-164.
- ↑ Erslev, A. J., and J. Caro. "Physiologic and molecular biology of erythropoietin." Medical oncology and tumor pharmacotherapy 3.3-4 (1986): 159-164.
- ↑ Erslev, A. J., and J. Caro. "Physiologic and molecular biology of erythropoietin." Medical oncology and tumor pharmacotherapy 3.3-4 (1986): 159-164.
- ↑ Jelkmann W. Erythropoietin after a century of research: younger than ever. Eur J Haematol. 2007 Mar;78(3):183-205. Epub 2007 Jan 23. PMID:17253966 doi:http://dx.doi.org/10.1111/j.1600-0609.2007.00818.x
- ↑ Constantinescu, Stefan N., Saghi Ghaffar, Harvey F. Lodish. The Erythropoietin Receptor: Structure, Activation and Intracellular Signal Transduction. Trends in Endocrinology and Metabolism, Volume10, Issue 1, 1January 1999, Pages 18-23.
- ↑ Constantinescu, Stefan N., Saghi Ghaffar, Harvey F. Lodish. The Erythropoietin Receptor: Structure, Activation and Intracellular Signal Transduction. Trends in Endocrinology and Metabolism, Volume10, Issue 1, 1January 1999, Pages 18-23.
- ↑ Constantinescu, Stefan N., Saghi Ghaffar, Harvey F. Lodish. The Erythropoietin Receptor: Structure, Activation and Intracellular Signal Transduction. Trends in Endocrinology and Metabolism, Volume10, Issue 1, 1January 1999, Pages 18-23.
- ↑ Syed RS, Reid SW, Li C, Cheetham JC, Aoki KH, Liu B, Zhan H, Osslund TD, Chirino AJ, Zhang J, Finer-Moore J, Elliott S, Sitney K, Katz BA, Matthews DJ, Wendoloski JJ, Egrie J, Stroud RM. Efficiency of signalling through cytokine receptors depends critically on receptor orientation. Nature. 1998 Oct 1;395(6701):511-6. PMID:9774108 doi:http://dx.doi.org/10.1038/26773
- ↑ Constantinescu, Stefan N., Saghi Ghaffar, Harvey F. Lodish. The Erythropoietin Receptor: Structure, Activation and Intracellular Signal Transduction. Trends in Endocrinology and Metabolism, Volume10, Issue 1, 1January 1999, Pages 18-23.