This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Amylase
From Proteopedia
(Difference between revisions)
| Line 4: | Line 4: | ||
=Structure<ref name="Main"/>= | =Structure<ref name="Main"/>= | ||
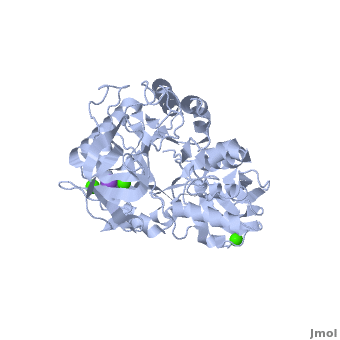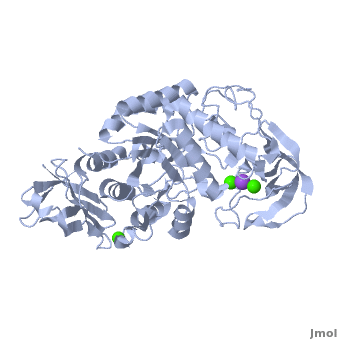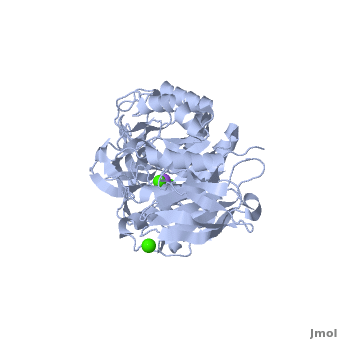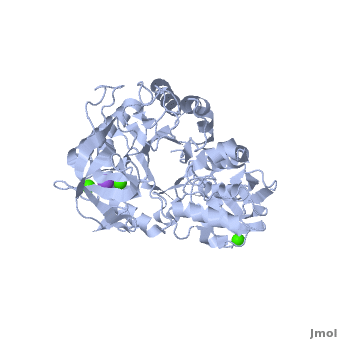
| - | Shown as 1hvx is the structure of the thermostable α-amylase of ''Bacillus stearothermophilus'' (BSTA)<ref name="Main"/>. BSTA is comprised of a single polypeptide chain. This chain is folded into three domains: A, B and C. These domains are generally found on all α-amylase enzymes. The <scene name='Sandbox_182/Domain_aa/1'>A domain </scene>constitutes the core structure, with a (β/α)<sub>8</sub>-barrel.The <scene name='Sandbox_182/Domain_a/1'> B domain</scene> consists of a sheet of four anti-parallel β-strands with a pair of anti-parallel β-strands. Long loops are observed between the β-strands. Located within the B domain is the <scene name='Sandbox_182/Trio/1'>binding site</scene> for Ca<sup>2+</sup>-Na<sup>+</sup>-Ca<sup>2+</sup>. <scene name='Sandbox_182/Domain_c/1'>Domain C </scene>consisting of eight β-strands is assembled into a globular unit forming a Greek key motif. It also holds the <scene name='Sandbox_182/Caiii/1'>third </scene>Ca<sup>2+</sup> binding site in association with domain A. Positioned on the C-terminal side of the β-strands of the (β/α)<sub>8</sub>-barrel in domain A is the active site. The catalytic residues involved for the BSTA active site are | + | Shown as 1hvx is the structure of the thermostable α-amylase of ''Bacillus stearothermophilus'' (BSTA)<ref name="Main"/>. BSTA is comprised of a single polypeptide chain. This chain is folded into three domains: A, B and C. These domains are generally found on all α-amylase enzymes. The <scene name='Sandbox_182/Domain_aa/1'>A domain </scene>constitutes the core structure, with a (β/α)<sub>8</sub>-barrel.The <scene name='Sandbox_182/Domain_a/1'> B domain</scene> consists of a sheet of four anti-parallel β-strands with a pair of anti-parallel β-strands. Long loops are observed between the β-strands. Located within the B domain is the <scene name='Sandbox_182/Trio/1'>binding site</scene> for Ca<sup>2+</sup>-Na<sup>+</sup>-Ca<sup>2+</sup>. <scene name='Sandbox_182/Domain_c/1'>Domain C </scene>consisting of eight β-strands is assembled into a globular unit forming a Greek key motif. It also holds the <scene name='Sandbox_182/Caiii/1'>third </scene>Ca<sup>2+</sup> binding site in association with domain A. Positioned on the C-terminal side of the β-strands of the (β/α)<sub>8</sub>-barrel in domain A is the active site. The catalytic residues involved for the BSTA active site are <scene name='38/382954/Active_site_ball_stick/2'>Asp234, Glu264, and Asp331</scene>. The residues are identical to other α-amylases, yet there are positional differences which reflect the flexible nature of catalytic resides. |
| - | <scene name=' | + | |
<scene name='Sandbox_182/Trio/1'>CaII and CaI with Na</scene> found in the interior of domain B and <scene name='Sandbox_182/Caiii/2'>CaIII </scene>at the interface of domain A and C, constitute the metal ion binding sites. All α-amylases contain one strongly conserved Ca<sup>2+</sup> ion for structural integrity and enzymatic activity.<ref name="chloride">PMID: 12021442</ref> CaI is consistent in α-amylases, however there are structural differences between the linear trio of CaI, CaII and Na in other enzymes. CaIII acts as a bridge between two loops, one from α6 of domain A, and one between β1 and β2 of domain C. | <scene name='Sandbox_182/Trio/1'>CaII and CaI with Na</scene> found in the interior of domain B and <scene name='Sandbox_182/Caiii/2'>CaIII </scene>at the interface of domain A and C, constitute the metal ion binding sites. All α-amylases contain one strongly conserved Ca<sup>2+</sup> ion for structural integrity and enzymatic activity.<ref name="chloride">PMID: 12021442</ref> CaI is consistent in α-amylases, however there are structural differences between the linear trio of CaI, CaII and Na in other enzymes. CaIII acts as a bridge between two loops, one from α6 of domain A, and one between β1 and β2 of domain C. | ||
==Chloride Dependent Enzymes== | ==Chloride Dependent Enzymes== | ||
Revision as of 01:50, 7 April 2015
| |||||||||||
3D structures of amylase
Updated on 07-April-2015
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 Yamamoto T.1988. Handbook of Amylases and Related Enzymes: Their Sources, Isolation Methods, Properties and Applications. Osaka Japan: Pergamon Press
- ↑ 2.0 2.1 Aghajari N, Feller G, Gerday C, Haser R. Crystal structures of the psychrophilic alpha-amylase from Alteromonas haloplanctis in its native form and complexed with an inhibitor. Protein Sci. 1998 Mar;7(3):564-72. PMID:9541387
- ↑ 3.0 3.1 3.2 Suvd D, Fujimoto Z, Takase K, Matsumura M, Mizuno H. Crystal structure of Bacillus stearothermophilus alpha-amylase: possible factors determining the thermostability. J Biochem. 2001 Mar;129(3):461-8. PMID:11226887
- ↑ 4.0 4.1 4.2 Aghajari N, Feller G, Gerday C, Haser R. Structural basis of alpha-amylase activation by chloride. Protein Sci. 2002 Jun;11(6):1435-41. PMID:12021442
- ↑ Maurus R, Begum A, Williams LK, Fredriksen JR, Zhang R, Withers SG, Brayer GD. Alternative catalytic anions differentially modulate human alpha-amylase activity and specificity(,). Biochemistry. 2008 Mar 18;47(11):3332-44. Epub 2008 Feb 20. PMID:18284212 doi:10.1021/bi701652t
- ↑ 6.0 6.1 Maurus R, Begum A, Williams LK, Fredriksen JR, Zhang R, Withers SG, Brayer GD. Alternative catalytic anions differentially modulate human alpha-amylase activity and specificity(,). Biochemistry. 2008 Mar 18;47(11):3332-44. Epub 2008 Feb 20. PMID:18284212 doi:10.1021/bi701652t
- ↑ 7.0 7.1 7.2 7.3 Kuriki T, Imanaka T. The concept of the alpha-amylase family: structural similarity and common catalytic mechanism. J Biosci Bioeng. 1999;87(5):557-65. PMID:16232518
- ↑ 8.0 8.1 PPMID: 17713601
- ↑ Franco OL, Rigden DJ, Melo FR, Grossi-De-Sa MF. Plant alpha-amylase inhibitors and their interaction with insect alpha-amylases. Eur J Biochem. 2002 Jan;269(2):397-412. PMID:11856298
- ↑ Yang RW, Shao ZX, Chen YY, Yin Z, Wang WJ. Lipase and pancreatic amylase activities in diagnosis of acute pancreatitis in patients with hyperamylasemia. Hepatobiliary Pancreat Dis Int. 2005 Nov;4(4):600-3. PMID:16286272
Proteopedia Page Contributors and Editors (what is this?)
Shane Riley, Michal Harel, Joel L. Sussman, Randi Woodbeck, Jaime Prilusky, Alexander Berchansky, Ann Taylor, Andrea Gorrell, David Canner