This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1058
From Proteopedia
(Difference between revisions)
| Line 15: | Line 15: | ||
Helix swapping is observed between two monomers to form stable dimers. The 11th and 12th helices of each monomer exchange three dimensional placement with the respective helices of the opposite monomer. Due to the 222 symmetry observed, only two dimers form than combine to form the observed tetramer. As a result of this structure, 18% of the surface of each monomer is buried within the protein. | Helix swapping is observed between two monomers to form stable dimers. The 11th and 12th helices of each monomer exchange three dimensional placement with the respective helices of the opposite monomer. Due to the 222 symmetry observed, only two dimers form than combine to form the observed tetramer. As a result of this structure, 18% of the surface of each monomer is buried within the protein. | ||
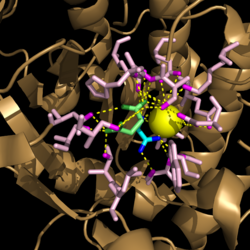
| + | ===Ligand Bound=== | ||
| + | [[Image:Active_Site_Hydrogen_Bonding.png|250 px|center|thumb|'''Figure 2. Active site residues hydrogen bound to a cofactor and the products of the catalyzed isocitrate reaction.''' Glyoxylate is shown in blue, succinate is shown in green, and the Mg<sup>2+</sup> cofactor is shown in yellow.]] | ||
== Function == | == Function == | ||
Revision as of 17:37, 3 April 2015
Isocitrate Lyase from Mycobacterium tuberculosis
| |||||||||||