This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1075
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
| - | ==Binding Specificity of EspG5 to PE25-PPE41 Proteins in '' | + | ==Binding Specificity of EspG5 to PE25-PPE41 Proteins in ''Mycobacterium tuberculosis''== |
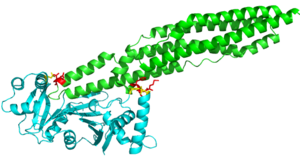
<StructureSection load='4KXR' size='340' side='right' caption='Here shows PE25-PPE41 ligand bound to EspG5 protein. Resolution 2.60Å' scene=''> | <StructureSection load='4KXR' size='340' side='right' caption='Here shows PE25-PPE41 ligand bound to EspG5 protein. Resolution 2.60Å' scene=''> | ||
| Line 33: | Line 33: | ||
<scene name='69/694242/Specific_contact_residues/1'>Random Loop, B2-B3 interactions</scene> | <scene name='69/694242/Specific_contact_residues/1'>Random Loop, B2-B3 interactions</scene> | ||
| - | The random loop on the cigar shaped PE-PPE ligand binds to the | + | The random loop on the cigar shaped PE-PPE ligand binds to the β2-β3 sheets on this EspG protein. |
The residues on the random turn in the PE-PPE protein are key for the specificity to the EspG protein. Coils between PE-PPE proteins vary greatly and influence binding affinity. Combined with its β-2 & β-3 interactions on the EspG protein, the EspG protein is PE-PPE specific. These make up the bulk of residue interactions in the complex. | The residues on the random turn in the PE-PPE protein are key for the specificity to the EspG protein. Coils between PE-PPE proteins vary greatly and influence binding affinity. Combined with its β-2 & β-3 interactions on the EspG protein, the EspG protein is PE-PPE specific. These make up the bulk of residue interactions in the complex. | ||
| Line 59: | Line 59: | ||
=== Pocket Residues === | === Pocket Residues === | ||
| - | EspG5 can bind to PE25-PPE41 due to a hand full of amino acid interactions. Most notably we have a | + | EspG5 can bind to PE25-PPE41 due to a hand full of amino acid interactions. Most notably we have a Pro51 on alpha-2 helix of the EspG5 protein. Also we have various contact residues on the random turn that interact with the B2-B3 sub unit, particularly the Glu127 of the random turn on the PE-PPE ligand. There are a few hydrophobic residues on the PE-PPE protein involved with binding affinity, the specific residues are Ala124, Leu125, Trp143, Gly147. |
Revision as of 01:11, 8 April 2015
Binding Specificity of EspG5 to PE25-PPE41 Proteins in Mycobacterium tuberculosis
| |||||||||||
References
- ↑ Ekiert DC, Cox JS. Structure of a PE-PPE-EspG complex from Mycobacterium tuberculosis reveals molecular specificity of ESX protein secretion. Proc Natl Acad Sci U S A. 2014 Oct 14;111(41):14758-63. doi:, 10.1073/pnas.1409345111. Epub 2014 Oct 1. PMID:25275011 doi:http://dx.doi.org/10.1073/pnas.1409345111