This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1066
From Proteopedia
(Difference between revisions)
| Line 18: | Line 18: | ||
== General mechanism for the activation of fatty acids == | == General mechanism for the activation of fatty acids == | ||
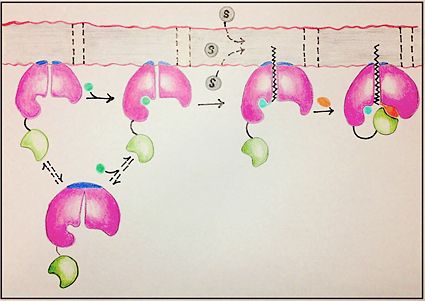
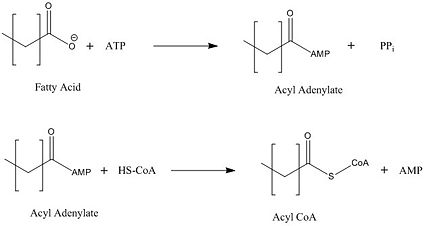
| - | FadD13 represents the first Fatty Acyl-CoA Synthetase of its kind to display biphasic kinetics.<ref name="residue paper"/> FadD13 first activates the fatty acid through a reaction with ATP to form an acyl adenylate intermediate and subsequently releases a pyrophosphate. Following a conformational change of the enzyme, coenzyme A is able to bind and | + | FadD13 represents the first Fatty Acyl-CoA Synthetase of its kind to display biphasic kinetics.<ref name="residue paper"/> FadD13 first activates the fatty acid through a reaction with ATP to form an acyl adenylate intermediate and subsequently releases a pyrophosphate. Following a conformational change of the enzyme upon the binding of ATP, coenzyme A is able to bind to its active site and react with the acyl adenylate intermediate forming the [http://en.wikipedia.org/wiki/Acyl-CoA acyl CoA] product (Figure 2). These activated fatty acyl-CoA thioesters have then been demonstrated to be important for the synthesis of triacyglycerols and phospholipids in the membrane of ''Mycobacterium tuberculosis''. <ref name="residue paper"/> |
Revision as of 02:36, 9 April 2015
| This Sandbox is Reserved from 02/09/2015, through 05/31/2016 for use in the course "CH462: Biochemistry 2" taught by Geoffrey C. Hoops at the Butler University. This reservation includes Sandbox Reserved 1051 through Sandbox Reserved 1080. |
To get started:
More help: Help:Editing |
Mycobacterium tuberculosis very-long-chain fatty acyl-CoA synthetase
| |||||||||||
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 Andersson CS, Lundgren CA, Magnusdottir A, Ge C, Wieslander A, Molina DM, Hogbom M. The Mycobacterium tuberculosis Very-Long-Chain Fatty Acyl-CoA Synthetase: Structural Basis for Housing Lipid Substrates Longer than the Enzyme. Structure. 2012 May 2. PMID:22560731 doi:10.1016/j.str.2012.03.012
- ↑ Jatana N, Jangid S, Khare G, Tyagi AK, Latha N. Molecular modeling studies of Fatty acyl-CoA synthetase (FadD13) from Mycobacterium tuberculosis--a potential target for the development of antitubercular drugs. J Mol Model. 2011 Feb;17(2):301-13. doi: 10.1007/s00894-010-0727-3. Epub 2010 May, 8. PMID:20454815 doi:http://dx.doi.org/10.1007/s00894-010-0727-3
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 Khare G, Gupta V, Gupta RK, Gupta R, Bhat R, Tyagi AK. Dissecting the role of critical residues and substrate preference of a Fatty Acyl-CoA Synthetase (FadD13) of Mycobacterium tuberculosis. PLoS One. 2009 Dec 21;4(12):e8387. doi: 10.1371/journal.pone.0008387. PMID:20027301 doi:10.1371/journal.pone.0008387
- ↑ 4.0 4.1 4.2 Jatana N, Jangid S, Khare G, Tyagi AK, Latha N. Molecular modeling studies of Fatty acyl-CoA synthetase (FadD13) from Mycobacterium tuberculosis--a potential target for the development of antitubercular drugs. J Mol Model. 2011 Feb;17(2):301-13. doi: 10.1007/s00894-010-0727-3. Epub 2010 May, 8. PMID:20454815 doi:http://dx.doi.org/10.1007/s00894-010-0727-3
External Resources
Tuberculosis Wikipedia page
Mycobacterium tuberculosis Wikipedia page
Coenzyme A Wikipedia page
Acyl CoA Wikipedia Page
Mycolic Acid Wikipedia page