This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1058
From Proteopedia
(Difference between revisions)
| Line 42: | Line 42: | ||
===Inhibitors=== | ===Inhibitors=== | ||
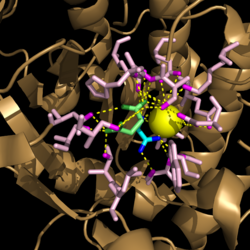
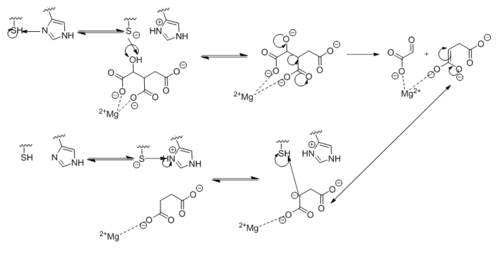
Due to the increased usefulness of this enzyme in propagating ''M. tuberculosis'' infections, specific inhibitors are being looked into as possible therapeutic targets for isocitrate lyase. Two such inhibitors that have already been identified are bromopyruvate and nitropropionate. Unfortunately, these molecules are non-specific and would also inhibit other enzymes essential for host function. <ref name="dunn"> Dunn, M.; Ramírez-Trujillo, J.; Hernández-Lucas, I.; Major roles of isocitrate lyase and malate synthase in bacterial and fungal pathogenesis. ''Microbiology''. '''2009'''. ''155'':3166-3175. doi:10.1099/mic.0.030858-0. </ref> More research is needed to identify inhibitors that selectively target enzymes in the glyoxylate cycle. | Due to the increased usefulness of this enzyme in propagating ''M. tuberculosis'' infections, specific inhibitors are being looked into as possible therapeutic targets for isocitrate lyase. Two such inhibitors that have already been identified are bromopyruvate and nitropropionate. Unfortunately, these molecules are non-specific and would also inhibit other enzymes essential for host function. <ref name="dunn"> Dunn, M.; Ramírez-Trujillo, J.; Hernández-Lucas, I.; Major roles of isocitrate lyase and malate synthase in bacterial and fungal pathogenesis. ''Microbiology''. '''2009'''. ''155'':3166-3175. doi:10.1099/mic.0.030858-0. </ref> More research is needed to identify inhibitors that selectively target enzymes in the glyoxylate cycle. | ||
| + | |||
| + | |||
| + | ==3D Structures of Isocitrate Lyase== | ||
| + | [http://www.rcsb.org/pdb/explore/explore.do?structureId=1DQU 1DQU]from ''Aspergillus nidulans''. | ||
Revision as of 03:08, 10 April 2015
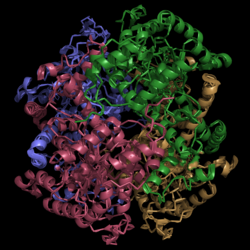
Isocitrate Lyase from Mycobacterium tuberculosis
| |||||||||||
References
- ↑ Cozzone, A.; Regulation of acetate metabolism by protein phosphorylation in enteric bacteria. Annual Review of Microbiology. 1998, 52:127-164. doi: 10.1146/annurev.micro.52.1.127.
- ↑ Srivastava, V.; Janin, A.; Srivastava, B.; Srivastava, R.; Selection of genes of Mycobacterium tuberculosis upregulated during residence in lungs of infected mice. ScienceDirect. 2007. doi:10.1016/j.tube.2007.10.002.
- ↑ Muñoz-Elías, E.; McKinney, J.; M. tuberculosis isocitrate lyases 1 and 2 are jointly required for in vivo growth and virulence. Nat. Med. 2005. 11(6):638-644. doi:10.1038/nm1252.
- ↑ Dunn, M.; Ramírez-Trujillo, J.; Hernández-Lucas, I.; Major roles of isocitrate lyase and malate synthase in bacterial and fungal pathogenesis. Microbiology. 2009. 155:3166-3175. doi:10.1099/mic.0.030858-0.