This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1072
From Proteopedia
(Difference between revisions)
| Line 13: | Line 13: | ||
| - | The two [http://en.wikipedia.org/wiki/Protein_domain domains] of each monomer are primarily [http://en.wikipedia.org/wiki/Alpha_helix alpha helical] and have similar foldings. The similar foldings suggests that the monomer results from a [http://en.wikipedia.org/wiki/Gene_duplication gene duplication] event; however, the C-terminal domain does not contain the [http://en.wikipedia.org/wiki/Heme_B heme ''b''] prosthetic group, while the <scene name='69/694238/N_terminus/ | + | The two [http://en.wikipedia.org/wiki/Protein_domain domains] of each monomer are primarily [http://en.wikipedia.org/wiki/Alpha_helix alpha helical] and have similar foldings. The similar foldings suggests that the monomer results from a [http://en.wikipedia.org/wiki/Gene_duplication gene duplication] event; however, the C-terminal domain does not contain the [http://en.wikipedia.org/wiki/Heme_B heme ''b''] prosthetic group, while the |
| + | <scene name='69/694238/N_terminus/5'>N terminal</scene> does. The [http://en.wikipedia.org/wiki/Active_site active site] is therefore located within the N-terminal domain. The two monomers interact through an interlocking hook formed by the N-terminal domains that stabilizes the formation of the dimer <ref name="one">PMID: 1523184</ref>. | ||
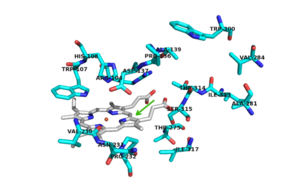
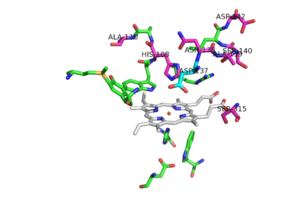
The N-terminal <scene name='69/694238/N_terminus/2'>hook</scene> is formed through hydrophobic interactions between residues Tyr-28 and Tyr-197 and residues Trp-38 and Trp-204. This interlocking loop region is also found in similar conformations of other catalase peroxidase structures such as: [http://www.proteopedia.org/wiki/index.php/1itk ''hm''CP] and ''bp''CP. | The N-terminal <scene name='69/694238/N_terminus/2'>hook</scene> is formed through hydrophobic interactions between residues Tyr-28 and Tyr-197 and residues Trp-38 and Trp-204. This interlocking loop region is also found in similar conformations of other catalase peroxidase structures such as: [http://www.proteopedia.org/wiki/index.php/1itk ''hm''CP] and ''bp''CP. | ||
| Line 28: | Line 29: | ||
<scene name='69/694238/Active_site/3'>Arg 104, Trp 107, His 108, His 270, Asp 381</scene> <ref name="one"/>. | <scene name='69/694238/Active_site/3'>Arg 104, Trp 107, His 108, His 270, Asp 381</scene> <ref name="one"/>. | ||
| - | <scene name='69/694238/N_terminus/5'>N terminus</scene> | ||
<scene name='69/694238/N_terminus/6'>N terminus stabilization with residues</scene> | <scene name='69/694238/N_terminus/6'>N terminus stabilization with residues</scene> | ||
Revision as of 16:25, 20 April 2015
| This Sandbox is Reserved from 02/09/2015, through 05/31/2016 for use in the course "CH462: Biochemistry 2" taught by Geoffrey C. Hoops at the Butler University. This reservation includes Sandbox Reserved 1051 through Sandbox Reserved 1080. |
To get started:
More help: Help:Editing |
| |||||||||||