This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1063
From Proteopedia
(Difference between revisions)
| Line 10: | Line 10: | ||
== Structural Highlights == | == Structural Highlights == | ||
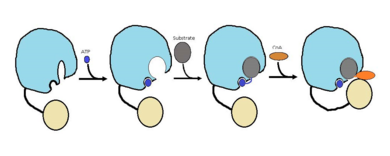
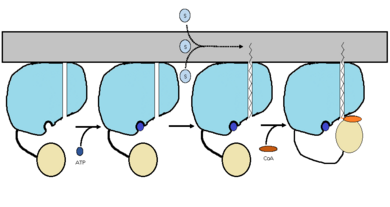
| - | FadD13 is composed of 503 amino acids which are divided into two domains. The larger of the two domains is the N-terminal domain composed of <scene name='69/694230/Fadd13_subunits/8'>residues 1-395</scene> shown in blue. The smaller of the two domains is the C-terminal domain composed of <scene name='69/694230/Fadd13_subunits/10'>residues 402-503</scene> shown in yellow. These two domains are held together by a flexible 6 amino acid linker (<scene name='69/694230/Fadd13_subunits/11'>residues 396-401</scene>) shown in black | + | FadD13 is composed of 503 amino acids which are divided into two domains. The larger of the two domains is the N-terminal domain composed of <scene name='69/694230/Fadd13_subunits/8'>residues 1-395</scene> shown in blue. The smaller of the two domains is the C-terminal domain composed of <scene name='69/694230/Fadd13_subunits/10'>residues 402-503</scene> shown in yellow. These two domains are held together by a flexible 6 amino acid linker (<scene name='69/694230/Fadd13_subunits/11'>residues 396-401</scene>) shown in black<ref name="Anderson 2012"/>. Research has also shown that altering V209D, D382A, and W377A effects the structual stability of FadD13. Val 209 and Asp 382 showed marginally reduced cytoplasmic expression, while Trp 377 showed a noteworthy low cytosolic expression. |
===Active Site=== | ===Active Site=== | ||
The active site of FadD13 is composed of an <scene name='69/694230/Fadd13_subunits/12'>ATP/AMP binding region</scene>. This region is comprised of residues 164-TSGTTGHPKG173-173 shown in red which binds to the phosphate group, and residues 298-VQGYALTE-305 shown in blue which binds to the adenine group. Research has also shown that <scene name='69/694230/Fadd13_subunits/16'>Ser 404</scene> plays a major role in binding of CoA. Ser 404 was shown to have a 4-fold enhancement for the Km value of CoA.<ref name="Khare 2009">DOI: 10.1371/journal.pone.0008387</ref> | The active site of FadD13 is composed of an <scene name='69/694230/Fadd13_subunits/12'>ATP/AMP binding region</scene>. This region is comprised of residues 164-TSGTTGHPKG173-173 shown in red which binds to the phosphate group, and residues 298-VQGYALTE-305 shown in blue which binds to the adenine group. Research has also shown that <scene name='69/694230/Fadd13_subunits/16'>Ser 404</scene> plays a major role in binding of CoA. Ser 404 was shown to have a 4-fold enhancement for the Km value of CoA.<ref name="Khare 2009">DOI: 10.1371/journal.pone.0008387</ref> | ||
Revision as of 18:38, 21 April 2015
FadD13
| |||||||||||
References
- ↑ Watkins PA, Maiguel D, Jia Z, Pevsner J. Evidence for 26 distinct acyl-coenzyme A synthetase genes in the human genome. J Lipid Res. 2007 Dec;48(12):2736-50. Epub 2007 Aug 30. PMID:17762044 doi:http://dx.doi.org/M700378-JLR200
- ↑ Kochan G, Pilka ES, von Delft F, Oppermann U, Yue WW. Structural snapshots for the conformation-dependent catalysis by human medium-chain acyl-coenzyme A synthetase ACSM2A. J Mol Biol. 2009 May 22;388(5):997-1008. Epub 2009 Apr 1. PMID:19345228 doi:10.1016/j.jmb.2009.03.064
- ↑ 3.0 3.1 3.2 3.3 3.4 Andersson CS, Lundgren CA, Magnusdottir A, Ge C, Wieslander A, Molina DM, Hogbom M. The Mycobacterium tuberculosis Very-Long-Chain Fatty Acyl-CoA Synthetase: Structural Basis for Housing Lipid Substrates Longer than the Enzyme. Structure. 2012 May 2. PMID:22560731 doi:10.1016/j.str.2012.03.012
- ↑ 4.0 4.1 Khare G, Gupta V, Gupta RK, Gupta R, Bhat R, Tyagi AK. Dissecting the role of critical residues and substrate preference of a Fatty Acyl-CoA Synthetase (FadD13) of Mycobacterium tuberculosis. PLoS One. 2009 Dec 21;4(12):e8387. doi: 10.1371/journal.pone.0008387. PMID:20027301 doi:10.1371/journal.pone.0008387